ANTACID REGULAR STRENGTH (calcium carbonate 500 mg) Dailymed
Generic: calcium carbonate 500mg is used for the treatment of Duodenal Ulcer Dyspepsia Esophagitis, Peptic Gastroesophageal Reflux Heartburn Gastrointestinal Hemorrhage Hypercalcemia Hypocalcemia Kidney Calculi Kidney Failure, Chronic Osteoporosis Stomach Ulcer Osteoporosis, Postmenopausal Hypophosphatemia
IMPRINT: GDC 126
SHAPE: round
COLOR: yellow
All Imprints
calcium carbonate 500 mg chewable tablet - gdc 126 round yellow
Go PRO for all pill images
Otc - Active Ingredient Section
Sennosides USP 15mg
Inactive Ingredient Section
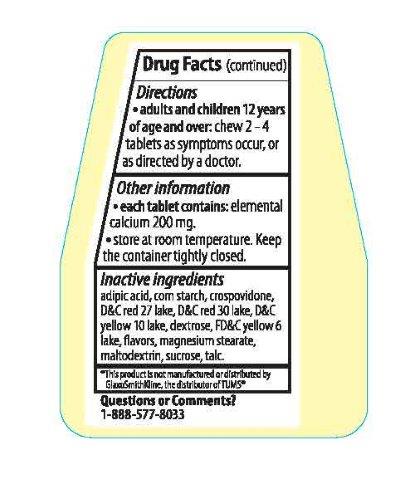
adipic acid, corn starch, crospovidone, D&C red 27 lake, D&C red 30 lake, D&C yellow 10 lake, dextrose, FD&C yellow 6 lake, flavors, magnesium stearate, maltodextrin, sucrose, talc.
Dosage & Administration Section
Adults and children 12 years of age and over: chew 2-4 tablets as symptoms occur, or as directed by a doctor.
Indications & Usage Section
Relieves:
- acid indigestion
- heartburn
Warnings Section
Ask a doctor or Pharmacist before use if you are presently taking a prescription drug. Antacids may interact with certain prescription drugs.
When using this product do not take more than 15 tablets in a 24-hour period, or use the maximum dosage of this product for more than 2 weeks, except under the advice and supervision of a doctor.
If pregnant or breast-feeding,ask a health professional before use.
Keep out of reach of children.
Otc - Keep Out Of Reach Of Children Section
Keep out of reach of children.
Otc - Purpose Section
Antacid
Other Safety Information
* Each tablet contains: elemental calcium 200mg.
* Store at room temperature. Keep the container tightly closed.
Package Label.principal Display Panel
DISCLAIMER:
"This tool does not provide medical advice, and is for informational and educational purposes only, and is not a substitute for professional medical advice, treatment or diagnosis. Call your doctor to receive medical advice. If you think you may have a medical emergency, please dial 911."
"Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. We may limit or otherwise restrict your access to the API in line with our Terms of Service."
"This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product."
PillSync may earn a commission via links on our site