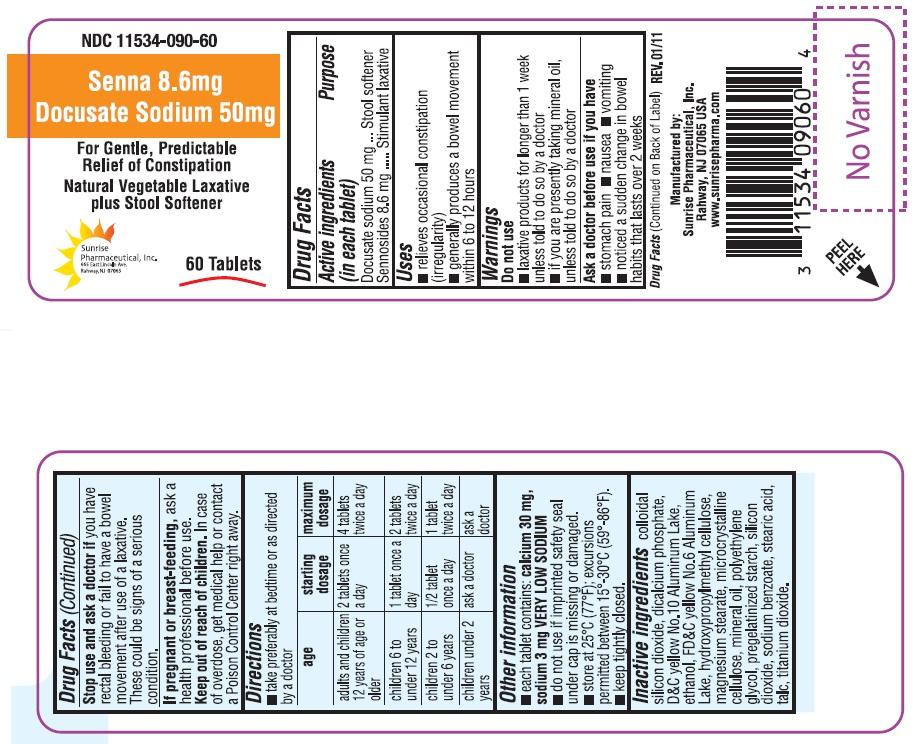
Senna and Docusate Sodium (docusate sodium 50 mg sennosides 8.6 mg) Dailymed
Generic: senna and docusate sodium is used for the treatment of Colic Constipation Intestinal Obstruction Nausea Vomiting Abdominal Pain
Go PRO for all pill images
Otc - Active Ingredient
Docusate sodium 50 mg
Sennosides 8.6 mg
Otc - Purpose
Stool softener and Stimulant laxative
Indications And Usage
- relieves occasional constipation (irregularity)
- generally produces a bowel movement within 6 to 12 hours
Warnings
Do not use:
- laxative products for longer than 1 week unless told to do so by a doctor
- if you are presently taking mineral oil, unless told to do so by a doctor
Otc - Ask A Doctor Before Use If You Have
- Stomach pain
- Nausea
- Vomiting
- Noticed a sudden change in bowel habits that lasts over 2 weeks
Otc - Stop Use And Ask A Doctor If
You have rectal bleeding or fail to have a bowel movement after use of a laxative. These could be signs of a serious condition.
Otc If Pregnant Or Breast Feeding
Ask a health professional before use.
Otc - Keep Out Of Reach Of Children
In case of overdose, get medical help or contact a Poison Control Center right away.
Dosage And Administration
- Take preferably at bedtime or as directed by a doctor
age starting dosage maximum dosage adults and children 12 years of age or older 2 tablets once a day 4 tablets twice a day children 6 to under 12 years 1 tablet once a day 2 tablets twice a day children 2 to under 6 years ½ tablet once a day 1 tablet twice a day children under 2 years ask a doctor ask a doctor
Other Information
- Each tablet contains: calcium 30 mg, sodium 3 mg VERY LOW SODIUM
- Do not use if imprinted safety seal under cap is missing or damaged.
- Store at 25° C (77° F); excursions permitted between 15° C-30° C (59° -86° F).
- Keep tightly closed.
Inactive Ingredient
Colloidal silicon dioxide, dicalcium phosphate, D&C yellow No 10 Aluminum Lake, ethanol, FD&C yellow No 6 Aluminum Lake, hydroxypropyl methyl cellulose, magnesium stearate, microcrystalline cellulose, mineral oil, polyethylene glycol, pregelatinized starch, silicon dioxide, sodium benzoate, stearic acid, talc, titanium dioxide
Package Label.principal Display Panel
DISCLAIMER:
"This tool does not provide medical advice, and is for informational and educational purposes only, and is not a substitute for professional medical advice, treatment or diagnosis. Call your doctor to receive medical advice. If you think you may have a medical emergency, please dial 911."
"Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. We may limit or otherwise restrict your access to the API in line with our Terms of Service."
"This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product."
PillSync may earn a commission via links on our site