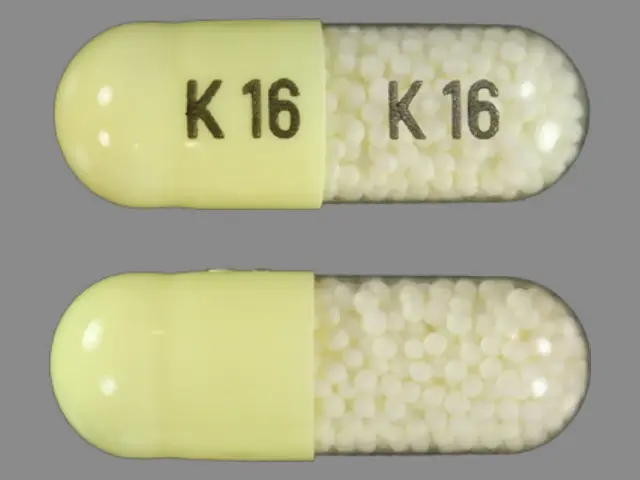
INDOMETHACIN (indomethacin 75 mg) Dailymed
Generic: indomethacin is used for the treatment of Arthritis, Rheumatoid Bursitis Ductus Arteriosus, Patent Gastrointestinal Hemorrhage Infant, Premature Inflammation Kidney Diseases Osteoarthritis Pain Peptic Ulcer Pregnancy Proctitis Spondylitis, Ankylosing Thrombocytopenia Arthritis, Gouty Enterocolitis, Necrotizing Tendinopathy
Boxed Warning
Boxed Warning Section
Go PRO for all pill images
Boxed Warning Section
Cardiovascular Thrombotic Events
• Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use (See and ). WARNINGS PRECAUTIONS
• Indomethacin Extended-release Capsules are contraindicated in the setting of coronary artery bypass graft (CABG) surgery (See and ). CONTRAINDICATIONS WARNINGS
Gastrointestinal Risk
• NSAIDs cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events. (See ). WARNINGS
Description:
Indomethacin Extended-release capsules USP 75 cannot be considered a simple analgesic and should not be used in conditions other than those recommended under . INDICATIONS AND USAGE
Indomethacin is a nonsteroidal, anti-inflammatory, indole derivative designated chemically as 1-(4-chlorobenzoyl)-5-methoxy-2-methyl-1 -indole-3-acetic acid. Indomethacin is practically insoluble in water and sparingly soluble in alcohol. It has a pKa of 4.5 and is stable in neutral or slightly acidic media and decomposes in strong alkali. The structural formula is: H

Each extended-release capsule, for oral administration contains 75 mg of indomethacin. In addition, each capsule contains the following inactive ingredients: corn starch, D&C Yellow# 10, gelatin, mannitol, povidone, sucrose, talc, and titanium dioxide.
This product meets USP Drug Release Test 2 Specifications.
Clinical Pharmacology:
Indomethacin is a nonsteroidal drug with anti-inflammatory, antipyretic and analgesic properties. Its mode of action, like that of other anti-inflammatory drugs, is not known. However, its therapeutic action is not due to pituitary-adrenal stimulation.
Indomethacin is a potent inhibitor of prostaglandin synthesis . Concentrations are reached during therapy which have been demonstrated to have an effect as well. Prostaglandins sensitize afferent nerves and potentiate the action of bradykinin in inducing pain in animal models. Moreover, prostaglandins are known to be among the mediators of inflammation. Since indomethacin is an inhibitor of prostaglandin synthesis, its mode of action may be due to a decrease of prostaglandins in peripheral tissues. in vitro in vivo
Indomethacin has been shown to be an effective anti-inflammatory agent, appropriate for long-term use in rheumatoid arthritis, ankylosing spondylitis, and osteoarthritis.
Indomethacin affords relief of symptoms; it does not alter the progressive course of the underlying disease.
Indomethacin suppresses inflammation in rheumatoid arthritis as demonstrated by relief of pain, and reduction of fever, swelling and tenderness. Improvement in patients treated with indomethacin for rheumatoid arthritis has been demonstrated by a reduction in joint swelling, average number of joints involved, and morning stiffness; by increased mobility as demonstrated by a decrease in walking time; and by improved functional capability as demonstrated by an increase in grip strength.
Indomethacin has been reported to diminish basal and CO stimulated cerebral blood flow in healthy volunteers following acute oral and intravenous administration. In one study, after one week of treatment with orally administered indomethacin, this effect on basal cerebral blood flow had disappeared. The clinical significance of this effect has not been established. 2
Indomethacin Extended-release Capsules USP 75 mg are designed to release 25 mg of drug initially and the remaining 50 mg over approximately 12 hours (90% of dose absorbed by 12 hours). Plasma concentrations of indomethacin fluctuate less and are more sustained following administration of Indomethacin Extended-release Capsules USP 75 mg than following administration of 25 mg indomethacin capsules given at 4 to 6 hour Intervals. In multiple-dose comparisons, the mean daily steady state plasma level of indomethacin attained with daily administration of Indomethacin Extended-release Capsules USP 75 mg was indistinguishable from that following indomethacin 25 mg capsules given at 0, 6, and 12 hours daily. However, there was a significant difference in indomethacin plasma levels between the two dosage regimens especially after 12 hours.
Controlled clinical studies of safety and efficacy in patients with osteoarthritis have shown that one capsule of Indomethacin Extended-release Capsules USP 75 mg was clinically comparable to one 25 mg indomethacin capsule t.i.d.; and in controlled clinical studies in patients with rheumatoid arthritis, one capsule of Indomethacin Extended-release Capsules USP 75 mg taken in the morning and one in the evening were clinically indistinguishable from one 50 mg capsule of indomethacin t.i.d.
Indomethacin is eliminated via renal excretion, metabolism, and biliary excretion. Indomethacin undergoes appreciable enterohepatic circulation. The mean half-life of indomethacin is estimated to be about 4.5 hours. With a typical therapeutic regimen of 25 or 50 mg t.i.d., the steady state plasma concentrations of indomethacin are an average 1.4 times those following the first dose.
Indomethacin exists in the plasma as the parent drug and its desmethyl, desbenzoyl, and desmethyl-desbenzoyl metabolites, all in the unconjugated form. About 60 percent of an oral dosage is recovered in urine as drug and metabolites (26 percent as indomethacin and its glucuronide), and 33 percent is recovered in feces (1.5 percent as indomethacin).
About 99% of indomethacin is bound to protein in plasma over the expected range of therapeutic plasma concentrations. Indomethacin has been found to cross the blood-brain barrier and the placenta.
Indications And Usage:
Carefully consider the potential benefits and risks of Indomethacin Extended-release Capsules USP 75 mg and other treatment options before deciding to use Indomethacin Extended-release Capsules USP 75 mg. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (See ). WARNINGS
Indomethacin Extended-release Capsules USP 75 mg has been found effective in active stages of the following:
1. Moderate to severe rheumatoid arthritis including acute flares of chronic disease.
2. Moderate to severe ankylosing spondylitis.
3. Moderate to severe osteoarthritis.
4. Acute painful shoulder (bursitis and/or tendinitis).
Indomethacin Extended-release Capsules USP 75 mg are not recommended for the treatment of acute gouty arthritis.
Indomethacin may enable the reduction of steroid dosage in patients receiving steroids for the more severe forms of rheumatoid arthritis. In such instances the steroid dosage should be reduced slowly and the patients followed very closely for any possible adverse effects.
The use of indomethacin in conjunction with aspirin or other salicylates is not recommended. Controlled clinical studies have shown that the combined use of indomethacin and aspirin does not produce any greater therapeutic effect than the use of indomethacin alone. Furthermore, in one of these clinical studies, the incidence of gastrointestinal side effects was significantly increased with combined therapy (See ). PRECAUTIONS, Drug Interactions
Contraindications:
Indomethacin Extended-release Capsules are contraindicated in patients with known hypersensitivity to Indomethacin.
Indomethacin Extended-release Capsules should not be given to patients who have experienced asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients (See , and ). WARNINGS PRECAUTIONS - Preexisting Asthma
In the setting of coronary artery bypass graft (CABG) surgery (See ). WARNINGS
Warnings:
Precautions:
General
Indomethacin Extended-release Capsules cannot be expected to substitute for corticosteroids or to treat corticosteroid insufficiency. Abrupt discontinuation of corticosteroids may lead to disease exacerbation. Patients on prolonged corticosteroid therapy should have their therapy tapered slowly if a decision is made to discontinue corticosteroids.
The pharmacological activity of Indomethacin Extended-release Capsules in reducing [fever and] inflammation may diminish the utility of these diagnostic signs in detecting complications of presumed noninfectious, painful conditions.
Borderline elevations of one or more liver tests may occur in up to 15% of patients taking NSAIDs including Indomethacin Extended-release Capsules. These laboratory abnormalities may progress, may remain unchanged, or may be transient with continuing therapy. Notable elevations of ALT or AST (approximately three or more times the upper limit of normal) have been reported in approximately 1% of patients in clinical trials with NSAIDs. In addition, rare cases of hepatic reactions, including jaundice and fatal fulminant hepatitis, liver necrosis and hepatic failure, some of them with fatal outcomes have been reported.
A patient with symptoms and/or signs suggesting liver dysfunction, or in whom an abnormal liver test has occurred, should be evaluated for evidence of the development of a more severe hepatic reaction while on therapy with Indomethacin Extended-release Capsules. If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, etc.), Indomethacin Extended-release Capsules should be discontinued.
Anemia is sometimes seen in patients receiving NSAIDs, including Indomethacin Extended-release Capsules. This may be due to fluid retention, occult or gross GI blood loss, or an incompletely described effect upon erythropoiesis. Patients on long-term treatment with NSAIDs, including Indomethacin Extended-release Capsules, should have their hemoglobin or hematocrit checked if they exhibit any signs or symptoms of anemia.
NSAIDs inhibit platelet aggregation and have been shown to prolong bleeding time in some patients. Unlike aspirin, their effect on platelet function is quantitatively less, of shorter duration, and reversible. Patients receiving Indomethacin Extended-release Capsules who may be adversely affected by alterations in platelet function, such as those with coagulation disorders or patients receiving anticoagulants, should be carefully monitored.
Patients with asthma may have aspirin-sensitive asthma. The use of aspirin in patients with aspirin-sensitive asthma has been associated with severe bronchospasm which can be fatal. Since cross reactivity, including bronchospasm, between aspirin and other nonsteroidal anti-inflammatory drugs has been reported in such aspirin-sensitive patients, Indomethacin Extended-release Capsules should not be administered to patients with this form of aspirin sensitivity and should be used with caution in patients with preexisting asthma.
Patients should be informed of the following information before initiating therapy with a NSAID and periodically during the course of ongoing therapy. Patients should also be encouraged to read the NSAID Medication Guide that accompanies each prescription dispensed.
1. Cardiovascular Thrombotic Events
Advise patients to be alert for the symptoms of cardiovascular thrombotic events, including chest pain, shortness of breath, weakness, or slurring of speech, and to report any of these symptoms to their health care provider immediately (See ). WARNINGS
2. Indomethacin Extended-release Capsules, like other NSAIDs, can cause GI discomfort and, rarely, serious GI side effects, such as ulcers and bleeding, which may result in hospitalization and even death. Although serious GI tract ulcerations and bleeding can occur without warning symptoms, patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing any indicative sign or symptoms including epigastric pain, dyspepsia, melena, and hematemesis. Patients should be apprised of the importance of this follow-up (See ). WARNINGS, Gastrointestinal Effects: Risk of Ulceration, Bleeding, and Perforation
3. Indomethacin Extended-release Capsules, like other NSAIDs, can cause serious skin side effects such as exfoliative dermatitis, SJS, and TEN, which may result in hospitalization and even death. Although serious skin reactions may occur without warning, patients should be alert for the signs and symptoms of skin rash and bulers, fever, or other signs of hypersensitivity such as itching, and should ask for medical advice when observing any indicative signs or symptoms. Patients should be advised to stop the drug immediately if they develop any type of rash and contact their physicians as soon as possible.
4. Heart Failure And Edema
Advise patients to be alert for the symptoms of congestive heart failure including shortness of breath, unexplained weight gain, or edema and to contact their healthcare provider if such symptoms occur (See ). WARNINGS
5. Patients should be informed of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If these occur, patients should be instructed to stop therapy and seek immediate medical therapy.
6. Patients should be informed of the signs of an anaphylactoid reaction (e.g., difficulty breathing, swelling of the face or throat). If these occur, patients should be instructed to seek immediate emergency help (See ). WARNINGS
7. In late pregnancy, as with other NSAIDs, Indomethacin Extended-release Capsules should be avoided because it will cause premature closure of the ductus arteriosus.
Because serious GI tract ulcerations and bleeding can occur without warning symptoms, physicians should monitor for signs or symptoms of GI bleeding. Patients on long-term treatment with NSAIDs, should have their CBC and a chemistry profile checked periodically. If clinical signs and symptoms consistent with liver or renal disease develop, systemic manifestations occur (e.g., eosinophilia, rash, etc.) or if abnormal liver tests persist or worsen, Indomethacin Extended-release Capsules should be discontinued.
Reports suggest that NSAIDs may diminish the antihypertensive effect of ACE-inhibitors. This interaction should be given consideration in patients taking NSAIDs concomitantly with ACE-inhibitors.
When Indomethacin Extended-release Capsules are administered with aspirin, its protein binding is reduced, although the clearance of free Indomethacin Extended-release Capsules is not altered. The clinical significance of this interaction is not known; however, as with other NSAIDs, concomitant administration of Indomethacin and aspirin is not generally recommended because of the potential of increased adverse effects.
Clinical studies, as well as post marketing observations, have shown that Indomethacin Extended-release Capsules can reduce the natriuretic effect-of furosemide and thiazides in some patients. This response has been attributed to inhibition of renal prostaglandin synthesis. During concomitant therapy with NSAIDs, the patient should be observed closely for signs of renal failure (See ), as well as to assure diuretic efficacy. WARNINGS, Renal Effects
NSAIDs have produced an elevation of plasma lithium levels and a reduction in renal lithium clearance. The mean minimum lithium concentration increased 15% and the renal clearance was decreased by approximately 20%. These effects have been attributed to inhibition of renal prostaglandin synthesis by the NSAID. Thus, when NSAIDs and lithium are administered concurrently, subjects should be observed carefully for signs of lithium toxicity.
NSAIDs have been reported to competitively inhibit methotrexate accumulation in rabbit kidney slices. This may indicate that they could enhance the toxicity of methotrexate. Caution should be used when NSAIDs are administered concomitantly with methotrexate.
The effects of warfarin and NSAIDs on GI bleeding are synergistic, such that users of both drugs together have a risk of serious GI bleeding higher than users of either drug alone.
Only if positive interactions have been observed. (See 201.57 (f)(4)(N))
Usually only if significant findings have been observed. (See 201.57 (f)(5))
Reproductive studies conducted in rats and rabbits have not demonstrated evidence of developmental abnormalities. However, animal reproduction studies are not always predictive of human response. There are no adequate and well-controlled studies in pregnant women.
Because of the known effects of nonsteroidal anti-inflammatory drugs on the fetal cardiovascular system (closure of ductus arteriosus), use during pregnancy (particularly late pregnancy) should be avoided.
In rat studies with NSAIDs, as with other drugs known to inhibit prostaglandin synthesis, an increased incidence of dystocia, delayed parturition, and decreased pup survival occurred. The effects of Indomethacin Extended-release Capsules on labor and delivery in pregnant women are unknown.
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human-milk and because of the potential for serious adverse reactions in nursing infants from Indomethacin Extended-release Capsules, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Safety and effectiveness in pediatric patients below the age of 14 years old have not been established.
As with any NSAIDs, caution should be exercised in treating the elderly (65 years and older).
Adverse Reactions:
The adverse reactions for indomethacin capsules uled in the following table have been arranged into two groups: 1) incidence greater than 1% and (2) incidence less than 1%. The incidence for group (1) was obtained from 33 double-blind controlled clinical trials reported in the literature (1,092 patients). The incidence for group (2) was based on reports in clinical trials, in the literature, and on voluntary reports since marketing. The probability of a causal relationship exists between indomethacin and these adverse reactions, some of which have been reported only rarely.
In controlled clinical trials, the incidence of adverse reactions to Indomethacin Extended-release Capsules USP 75 mg and equal 24-hour doses of indomethacin capsules were similar.
Incidence great than 1% Incidence less than 1%
GASTROINTESTINAL
nausea* with or without vomiting
dyspepsia* (including indigestion, heartburn and epigastric pain)
diarrhea
abdominal distress or pain constipation                     Â
anorexia
bloating (includes distention)
flatulence
peptic ulcer
gastroenteritis
rectal bleeding
proctitis
single or multiple ulcerations, including perforation and hemorrhage of the esophagus, stomach,duodenum or small and large intestines
Intestinal ulceration associated with stenosis and obstruction
gastrointestinal bleeding without obvious ulcer formation and perforation of pre-existing sigmoid lesions (diverticulum, carcinoma, etc.) development of ulcerative colitis and regional ileitisulcerative stomatitis
toxic hepatitis and jaundice (some fatal cases have been  reported)
CENTRAL NERVOUS SYSTEM
headache (11.7%)
dizziness*Â
vertigo
somnolenceÂ
depression and fatigue
including malaise and ullessness)Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â Â
anxiety (includes nervousness)
muscle weakness
involuntary muscle movemets insomnia
muzziness
psychic disturbances including psychotic episodes
mental confussion                 Â
drowsiness                              Â
light-headedness
syncope
paresthesia
aggravation of epilepsy and parkinsonism
depersonalization
coma
peripheral neuropathy convulsions dysarthria                                       Â
SPECIAL SENSES
tinnitus                                  ocular-corneal deposits and retinal disturbances, including those of macula, have been reported in some patients on prolonged therapy with indomethacin                          Â
blurred vision
diplopia
hearing disturbances, deafness                                         Â
CARDIOVASCULAR
none                                    Â
hypertension
hypotension
tachycardia
chest pain                                       Â
congestive heart failure
arrhythmia; palpitations                                      Â
METABOLIC
none                                    Â
edema
weight gain
fluid retention
flushing or sweating                                    Â
hyperglycemia
glycosuria hyperkalemia                                   Â
INTEGUMENTARY
none                                    Â
pruritus
rash; urticaria
petechiae or ecchymosis                               Â
exfoliative dermatitis
erythema nodosum
loss of hair
Stevens-Johnson syndrome
erythema multiforme
toxic epidermal necrolysis                                         Â
HEMATOLOGIC
none                                   Â
leukopenia
bone marrow depression
anemia secondary to obvious or occult gastrointestinal
bleeding                                 Â
aplastic anemia
hemolytic anemia
agranulocytosis
thrombocytopenic
purpura
disseminated
intravascularÂ
coagulation                                       Â
HYPERSENSITIVITY
none                                          Â
acute anaphylaxis
acute respiratory
distress
rapid fall in blood
pressure resembling a shock
-like state angioedema                            Â
dyspnea
asthma
purpura
angiitis
pulmonary edema
fever                                                 Â
GENITOURINARY
none                                  Â
hematuria
vaginal bleeding
proteinuria
nephrotic syndrome
interstitial nephritis                                 Â
BUN elevation
renal insufficiencyÂ
including renal failure                                               Â
MISCELLANEOUS
none                                     Â
epistaxis
breast changes,
including enlargement and Â
tenderness, or gynecomastia                                                                                   Â
*Â Reactions occurring in 3% to 9% of patients treated with indomethacin. (Those reactions occurring in less than 3% of the patients are unmarked.)
Other reactions have been reported but occurred under circumstances where a causal relationship could not be established. However, in these rarely reported events, the possibility cannot be excluded. Therefore, these observations are being uled to serve as alerting information to physicians: Causal Relationship Unknown:
A rare occurrence of fulminant necrotizing fasciitis, particularly in association with Group A β-hemolytic streptococcus, has been described in persons treated with nonsteroidal anti-inflammatory agents, including indomethacin, sometimes with fatal outcome (See also ). PRECAUTIONS,General
: Thrombophlebitis. Cardiovascular
: Although there have been several reports of leukemia, the supporting information is weak. Hematologic
: Urinary frequency. Genitourinary
Overdosage:
The following symptoms may be observed following overdosage: nausea, vomiting, intense headache, dizziness, mental confusion, disorientation, or lethargy. There have been reports of paresthesias, numbness, and convulsions.
Treatment is symptomatic and supportive. The stomach should be emptied as quickly as possible if the ingestion is recent. If vomiting has not occurred spontaneously, the patient should be induced to vomit with syrup of ipecac. If the patient is unable to vomit, gastric lavage should be performed. Once the stomach has been emptied, 25 or 50 g of activated charcoal may be given. Depending on the condition of the patient, close medical observation and nursing care may be required. The patient should be followed for several days because gastrointestinal ulceration and hemorrhage have been reported as adverse reactions of indomethacin. Use of antacids may be helpful.
The oral LD of indomethacin in mice and rats (based on 14 day mortality response) was 50 and 12 mg/kg, respectively. 50
Dosage And Administration:
Carefully consider the potential benefits and risks of Indomethacin Extended-release Capsules and other treatment options before deciding to use Indomethacin Extended-release Capsules. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (See ). WARNINGS
Indomethacin Extended-release Capsules USP 75 mg are available for oral use. Indomethacin Extended-release Capsules USP 75 mg can be administered once a day and can be substituted for indomethacin 25 mg capsules t.i.d. However, there will be significant difference between the two dosage regimens in indomethacin blood levels, especially after 12 hours (See ). In addition, Indomethacin Extended-release Capsules USP 75 mg b.i.d. can be substituted for indomethacin 50 mg capsules t.i.d. Indomethacin Extended-release Capsules USP 75 mg may be substituted for all the indications of indomethacin capsules except acute gouty arthritis. CLINICAL PHARMACOLOGY
Adverse reactions appear to correlate with the size of the dose of indomethacin in most patients, but not all. Therefore, every effort should be made to determine the smallest effective dosage for the individual patient.
Always give Indomethacin Extended-release Capsules USP 75 mg with food, immediately after meals, or with antacids to reduce gastric irritation.
Indomethacin Extended-release Capsules USP 75 mg ordinarily should not be prescribed for children 14 years of age and under (See ). Pediatric Use: WARNINGS
Dosage Recommendations for Active Stages of the Following: Adult Use:
1.    Moderate to severe rheumatoid arthritis, including acute flares of chronic disesase; moderate to severe ankylosing spondylitis; and moderate to severe osteoarthritis.
The following information is provided as background only and refers to immediate-release indomethacin capsules (25 mg or 50 mg):Â
Suggested Dosage:
The following recommendations on dosing pertain to immediate-release indomethacin capsules USP, and provide important information regarding the dosage and administration of indomethacin. The prescriber should be aware of this information when considering and prescribing Indomethacin Extended-release Capsules USP 75 mg.
Indomethacin capsules 25 mg b.i.d. or t.i.d. If this is well tolerated, increase the daily dosage by 25 or 50 mg, if required by continuing symptoms, at weekly intervals until a satisfactory response is obtained or until a total daily dose of 150-200 mg is reached. DOSES ABOVE THIS AMOUNT GENERALLY DO NOT INCREASE THE EFFECTIVENESS OF THE DRUG.
In patients who have persistent night pain and/or morning stiffness, the giving of a large portion, up to a maximum of 100 mg, of the total daily dose at bedtime, either orally or by rectal suppositories, may be helpful in affording relief. The total daily dose should not exceed 200 mg. In acute flares of chronic rheumatoid arthritis, it may be necessary to increase the dosage by 25 mg or, if required, by 50 mg daily.
The following information refers to Indomethacin Extended-release Capsules USP 75 mg:
If Indomethacin Extended-release Capsules USP 75 mg are used for initiating indomethacin treatment, one capsule daily should be the usual starting dose in order to observe patient tolerance since 75 mg per day is the maximum recommended starting dose for indomethacin (see above). If Indomethacin Extended-release Capsules USP 75 mg are used to increase the daily dose, patients should be observed for possible signs and symptoms of intolerance since the daily increment will exceed the daily increment recommended for other dosage forms. For patients who require 150 mg of indomethacin per day and have demonstrated acceptable tolerance, Indomethacin Extended-release Capsules USP 75 mg may be prescribed as one capsule twice daily.
If minor adverse effects develop as the dosage is increased, reduce the dosage rapidly to a tolerated dose and OBSERVE THE PATIENT CLOSELY.
If severe adverse reactions occur, STOP THE DRUG. After the acute phase of the disease is under control, an attempt to reduce the daily dose should be made repeatedly until the patient is receiving the smallest effective dose or the drug is discontinued.
Careful instructions to, and observations of, the individual patient are essential to the prevention of serious, irreversible, including fatal, adverse reactions.
As advancing years appear to increase the possibility of adverse reactions, Indomethacin Extended-release Capsules should be used with greater care in the aged.
2. Acute painful shoulder (bursitis and/or tendinitis): Initial Dose: 75 mg to 150mg daily. When 150 mg is prescribed, give as one capsule twice daily.
The drug should be discontinued after the signs and symptoms of inflammation have been controlled for several days. The usual course of therapy is 7 to 14 days.
How Supplied:
NDC:68151-1733-8 in a BOTTLE of 1 CAPSULE, EXTENDED RELEASES
Medication Guide For Nonsteroidal Anti-inflammatory Drugs (nsaids)
What is the most important information I should know about medicines called Non-steroidal Anti-inflammatory Drugs (NSAIDs)?
NSAIDs can cause serious side effects, including:
• This risk may happen early in treatment and may increase: Increased risk of a heart attack or stroke that can lead to death.
° with increasing doses of NSAIDs
° with longer use of NSAIDs
Do not take NSAIDs right before or after a heart surgery called a "coronary artery bypass graft (CABG)’’.
Avoid taking NSAIDs after a recent heart attack, unless your healthcare provider tells you to. You may have an increased risk of another heart attack if you take NSAIDs after a recent heart attack.
• Increased risk of bleeding, ulcers, and tears (perforation) of the esophagus (tube leading from the mouth to the stomach), stomach and intestines:
° anytime during use
° without warning symptoms
° that may cause death
The risk of getting an ulcer or bleeding increases with:
° past history of stomach ulcers, or stomach or intestinal bleeding with use of NSAIDs
° taking medicines called "corticosteroids", "anticoagulants", "SSRIs", or "SNRIs"
° increasing doses of NSAIDs
° longer use of NSAIDs
° smoking
° drinking alcohol
° older age
° poor health
° advanced liver disease
° bleeding problems
NSAIDs should only be used:
° exactly as prescribed
° at the lowest dose possible for your treatment
° for the shortest time needed
What are NSAIDs?
NSAIDs are used to treat pain and redness, swelling, and heat (inflammation) from medical conditions such as different types of arthritis, menstrual cramps, and other types of short-term pain.
Who should not take NSAIDs?
Do not take NSAIDs:
• if you have had an asthma attack, hives, or other allergic reaction with aspirin or any other NSAIDs.
• right before or after heart bypass surgery.
Before taking NSAIDS, tell your healthcare provider about all of your medical conditions, including if you:
•have liver or kidney problems
• have high blood pressure
• have asthma
• are pregnant or plan to become pregnant. Talk to your healthcare provider if you are considering taking NSAIDs during pregnancy. You should not take NSAIDs after 29 weeks of pregnancy.
• are breastfeeding or plan to breast feed.
NSAIDs and some other medicines can interact with each other and cause serious side effects. Tell your healthcare provider about all of the medicines you take, including prescription or over-the-counter medicines, vitamins or herbal supplements. Do not start taking any new medicine without talking to your healthcare provider first.
What are the possible side effects of NSAIDs?
NSAIDs can cause serious side effects, including:
See "What is the most important information I should know about medicines called Nonsteroidal Anti-inflammatory Drugs (NSAIDs)"?
• new or worse high blood pressure
• heart failure
• liver problems including liver failure
• kidney problems including kidney failure
• low red blood cells (anemia)
• life-threatening skin reactions
• life threatening allergic reactions
• stomach pain, constipation, diarrhea, gas, heartburn, nausea, vomiting, and dizziness. Other side effects of NSAIDs include:
Get emergency help right away if you get any of the following symptoms:
•shortness of breath or trouble breathing
• slurred speech
• chest pain
• swelling of the face or throat
• weakness in one part or side of your body
Stop taking your NSAID and call your healthcare provider right away if you get any of the following symptoms :
• nausea
• vomit blood
• more tired or weaker than usual
• there is blood in your bowel movement or it is black and sticky like tar
• itching
• diarrhea
• unusual weight gain
• your skin or eyes look yellow
• skin rash or bulers with fever
• indigestion or stomach pain
• swelling of the arms and legs, hands and feet
• flu-like symptoms
If you take too much of your NSAID, call your healthcare provider or get medical help right away.
These are not all the possible side effects with NSAIDs. For more information, ask your healthcare provider or pharmacist about NSAIDs.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Other information about NSAIDs
• Aspirin is an NSAID but it does not increase the chance of a heart attack. Aspirin can cause bleeding in the brain, stomach, and intestines. Aspirin can also cause ulcers in the stomach and intestines.
• Some NSAIDs are sold in lower doses without a prescription (over-the-counter). Talk to your healthcare provider before using over-the-counter NSAIDs for more than 10 days.
General information about the safe and effective use of NSAIDs
Medicines are sometimes prescribed for purposes other than those uled in a Medication Guide. Do not use NSAIDs for a condition for which it was not prescribed. Do not give NSAIDs to other people, even if they have the same symptoms that you have. It may harm them.
If you would like more information about NSAIDs, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about NSAIDs that is written for health professionals.
Manufactured by:
KVK-TECH INC.
110 Terry Drive
Newtown, PA 18940

Item ID # 006030/06
Manufacturer’s Code: 10702       07/15
For more information, go to or call our customer service at 215-579-1842. www.kvktech.com
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Indomethacin Capsule, Extended Release
DISCLAIMER:
"This tool does not provide medical advice, and is for informational and educational purposes only, and is not a substitute for professional medical advice, treatment or diagnosis. Call your doctor to receive medical advice. If you think you may have a medical emergency, please dial 911."
"Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. We may limit or otherwise restrict your access to the API in line with our Terms of Service."
"This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product."
PillSync may earn a commission via links on our site