Ludent (sodium fluoride 0.25 mg) Dailymed
Generic: sodium fluoride is used for the treatment of Dental Caries Dental Plaque Osteoporosis Tooth Loss
Go PRO for all pill images
This insert is for 1.0 mg strength tablets, 0.5 mg strength tablets, and 0.25 mg strength tablets.
Rx Only
Description Section
This product is a prescription product for the clinical dietary management of the metabolic processes of caries prophylaxis.
7, 60 and120 Tablets per bottle and 30 count buler
Water FÂŻ Content Ages 0 ppm FÂŻ to <0.3 ppm FÂŻ 0.3 ppm FÂŻ to 0.6ppm FÂŻ >0.6ppm FÂŻ 3 yrs. to 6 yrs. 0.5mg Per day 0.25mg 0 >6yrs. to 16 yrs. 1.0mg 0.5mg 0
Active Ingredient: Fluoride (as Sodium Fluoride) 0.25 mg / 0.5 mg / 1.0 mg
Inactive Ingredients: Xylitol, microcrystalline cellulose, malic acid, magnesium stearate, talc, citric acid, orange flavor, sucralose.
Caution: Do not eat or drink dairy products within one hour of fluoride administration. Incompatibility of fluoride with dairy foods has been reported due to formation of calcium fluoride which is poorly absorbed.
Clinical Pharmacology
Sodium Fluoride acts systemically (before tooth eruption) and topically (post-eruption) by increasing tooth resistance to acid dissolution, by promoting remineralization, and by inhibiting the cariogenic microbial process.
Indications And Usage
For once daily self-applied systemic use as a dental caries preventive in pediatric patients. It has been established that ingestion of fluoridated drinking water (1 ppm FÂŻ) during the period of tooth development results in a significant decrease in the incidence of dental caries. Sodium Fluoride Chewable Tablets were developed to provide systemic fluoride for use as a supplement in pediatric patients from age 3 years to age 16 years and older living in areas where the drinking water fluoride contents does not exceed 0.6 ppm FÂŻ.
Contraindications
Fluoride 1.0 mg Tablets are contraindicated when the fluoride content of drinking water is >0.3 ppm FÂŻ and should not be administered to pediatric patients under 6 years. Fluoride 0.5 mg Tablets are contraindicated when the fluoride content of drinking water is more than 0.6 ppm FÂŻ and should not be administered to pediatric patients under age 6 when the fluoride content of drinking water is 0.3 ppm FÂŻ or more or to pediatric patients under age 3 years. Fluoride 0.25 mg Tablets are contraindicated when the fluoride content of drinking water is more than 0.6 ppm FÂŻ and should not be administered to pediatric patients under age 3 years when the fluoride content of drinking water is 0.3 ppm FÂŻ or more. Do not administer Sodium Fluoride Chewable Tablets (any strength) to pediatric patients under age 3 years. Sodium Fluoride Chewable Tablets (any strength) are not indicated for use in adults.
Overdosage
Prolonged daily ingestion of excessive fluoride will result in varying degrees of fluorisis. Accidental ingestion of large amounts of fluoride may result in acute burning in the mouth and sore tongue. Nausea, vomiting, and diarrhea may occur soon after ingestion (within 30 minutes) and are accompanied by salivation, hematemesis, and epigastric cramping abdominal pain. These symptoms may persist for 24 hours. If less than 5 mg fluoride/kg body weight (i.e., less than 2.3 mg fluoride/lb body weight) have been ingested, give calcium (e.g., milk) orally to relieve gastrointestinal symptoms and observe for a few hours. If more than 5 mg fluoride/kg body weight (i.e., more than 2.3 mg fluoride/lb body weight) have been ingested, induce vomiting, give orally soluble calcium (e.g., milk, 5% calcium gluconate or calcium lactate solution) and immediately seek medical assistance. For accidental ingestion of more than 15 mg fluoride/kg body weight (i.e., more than 6.9 mg fluoride/lb body weight), induce vomiting and admit immediately to a hospital facility. A treatment dose of Sodium Fluoride Chewable Tablets contains 0.25, 0.5 or 1 mg fluoride. The treatment of choice depends upon the age of the child and the water fluoride content. A bottle of 120 0.25 mg tablets contains 30 mg fluoride. A bottle of 120 0.5 mg tablets contains 60 mg fluoride. A bottle of 120 1 mg tablets contains 120 mg fluoride. [The total amount of sodium fluoride in a bottle of 120 Fluoride Chewable Tablets (all strengths) conforms with the recommended amount of the American Dental Association for the maximum to be dispensed at one time for safety purposes.]
Warning
Prolonged daily ingestion of quantities greater than the recommended amount may result in various degrees of dental fluorosis in pediatric patients under age 6 years, especially if the water fluoridation exceeds 0.6 ppm. Read directions carefully before using. This product, as all chewable tablets, is not recommended for children under age 3 due to risk of choking. Keep out of the reach of infants and children.
Precautions
General
Please refer to the CONTRAINDICATIONS, WARNINGS and OVERDOSAGE sections for overdosage concerns. Use in pediatric patients below the age of 3 years is not recommended by current American Dental Associations and American Academy of Pediatrics guidelines.
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a study conducted in rodents, no carcinogenesis was found in male and female rats treated with fluoride at dose levels ranging from 4.1 to 9.1 mg/kg of body weight. Equivocal evidence of carcinogenesis was reported for male rats treated with 2.5 and 4.1 mg/kg of body weight. In a second study, no carcinogenesis was observed in rats, males or females, treated with fluoride up to 11.3 mg/kg of body weight. This dose is at least 400 times greater than the recommended daily dose of Sodium Fluoride Chewable Tablets. Fluoride ion is not mutagenic in standard bacterial systems. It has been shown that fluoride ion has potential to induce chromosome aberrations in cultured human and rodent cells at does much higher than those to which humans are exposed. In vivo data is conflicting. Some studies report chromosome damage in rodents while other studies using similar protocols report negative results. Potential adverse reproductive effects of fluoride exposure in humans has not been adequately evaluated. Adverse effects on reproduction were reported for rats, mice, fox, and cattle exposed to 100 ppm or greater concentrations of fluoride in their diet or drinking water. Other studies conducted in rats demonstrated that lower doses of fluoride (5 mg/kg of body weight) did not result in impaired fertility and reproductive capabilities. This dose is approximately 200 times greater than the recommended daily dose of Sodium Fluoride Chewable Tablets.
Pregnancy
Teratogenic Effects
Pregnancy Category B
It has been shown that fluoride crosses the placenta of rats, but only 0.01% of the amount administered is incorporated in fetal tissue. Animal studies (rats, mice, rabbits) have shown that fluoride is not a teratogen. Maternal exposure to 12.2 mg fluoride/kg of body weight (rats) or 13.1 mg/kg of body weight (rabbits) did not affect the litter size or fetal weight and did not increase the frequency of skeletal or visceral malformations. Epidemiological studies conducted in areas with high levels of naturally fluoridated water showed no increase in birth defects. Heavy exposure to fluoride during in utero development may result in skeletal fluorosis which becomes evident in childhood.
Nursing Mothers
It is not known if fluoride is excreted in human milk. However, many drugs are excreted in human milk and caution should be exercised when Sodium Fluoride Chewable Tablets are administered to a nursing woman. Reduced milk production was reported in farm raised fox when the animals were fed a diet containing a high concentration of fluoride (98-137 mg/kg of body weight). No adverse effects on parturition, lactation, or offspring were seen in rats administered fluoride up to 5 mg/kg of body weight. This dose is at least 200 times greater than the recommended daily dose of Sodium Fluoride Chewable Tablets.
Geriatric Use
Sodium Fluoride Chewable Tablets (any strength) are not indicated for use in geriatric patients.
Pediatric Use
The use of Sodium Fluoride Chewable Tablets as a caries preventive in pediatric age groups 3 years to 16 years is supported by evidence from adequate and well controlled studies on fluoride supplementation from birth through adolescence.
Adverse Reactions
Allergic rash and other idiosyncrasies have been rarely reported.
Dosage And Administration
(for dosage see table)
Dissolve in the mouth or chew before swallowing, preferably at bedtime after brushing teeth. See schedule below to determine dosage.
How Supplied
Bottle of 7, 60 and 120 Chewable Tablets and 30 Count Blister
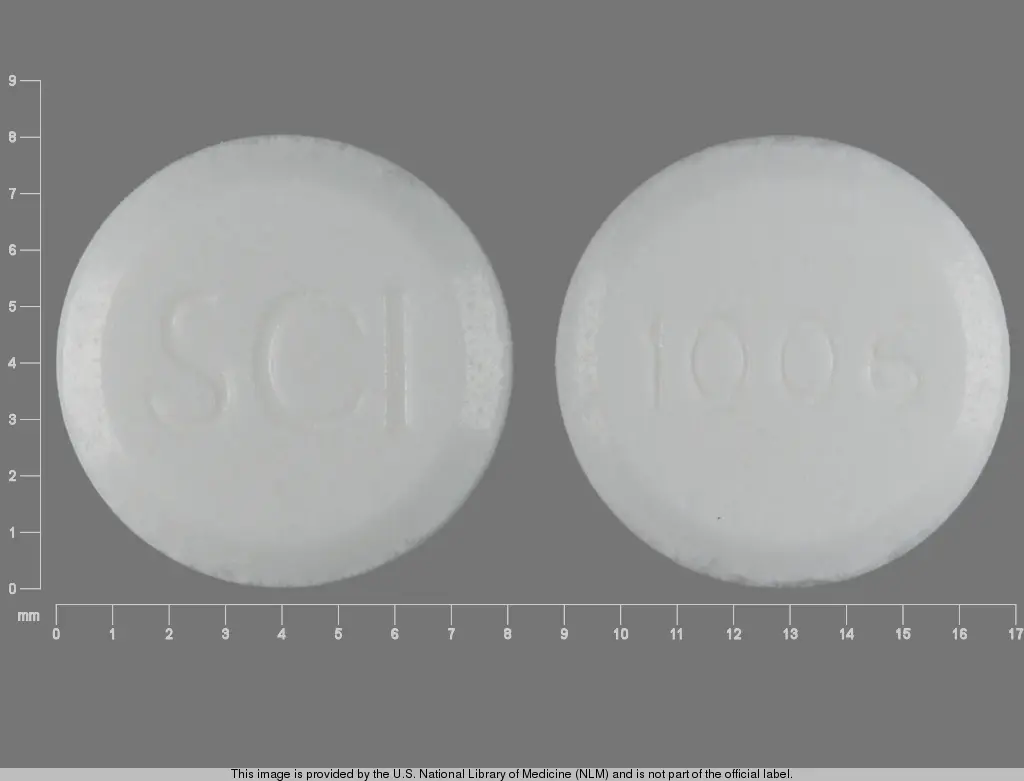
Each tablet contains 0.25 mg Fluoride from 0.55 mg Sodium Fluoride. White to off-white, orange flavor, round-shaped chewable tablet. Debossed "SCI" on one side and 1006 on the other. NDC 44946-1015-2 (60 count) , NDC 44946-1015-3 (120 count), NDC 44946-1015-6 (30 count buler).
Each tablet contains 0.5 mg Fluoride from 1.1 mg Sodium Fluoride. White to off-white, orange flavor, round-shaped chewable tablet. Debossed "SCI" on one side and 1007 on the other. NDC 44946-1016-7 (7 count sample size), NDC 44946-1016-2 (60 count), NDC 44946-1016-3 (120 count), NDC 44946-1016-6 (30 count buler).
Each tablet contains 1.0 mg Fluoride from 2.2 mg Sodium Fluoride. White to off-white, orange flavor, round-shaped chewable tablet. Debossed "SCI" on one side and 1004 on the other. NDC 44946-1017-2 (60 count) , NDC 44946-1017-3 (120 count), NDC 44946-1017-6 (30 count buler).
STORAGE
Store at 25° C (77° F); excursions permitted to 15° - 30° C (59° - 86° F). See USP Controlled Room Temperature. Protect from light and moisture.
References
- Accepted Dental Therapeutics, Ed. 40, American Dental Association, Chicago, 399-402 (1984).
- J. Jakush, New Fluoride Schedule Adopted, ADA News, 12, 14 (May 16, 1994).
- Aasenden, R., and Peebles, T.C. "Effects of Fluoride Supplementation From Birth on Dental Caries and Fluorisis in Teenaged Children", Arch. Oral, Biol., 23, 111–115 (1974).
- Hamber, L. "Controlled Trial of Fluoride Vitamin Drops for Prevention of Caries in Children", Lancet, 1, 441-442 (1971).
- Hennon, D.K. Stookey, G.K. and Veiswanter, B.B. "Fluoride-Vitamin Supplements: Effects on Dental Caries and Fluorisis When Used in Areas with Suboptimum Fluoride in the Water Supply", JADA, 95, 965-971 (1977).
Dispense in a tight, light-resistant container with a child-resistant closure as defined in the USP/NF.
Warning: Keep this and all medications out of the reach of children. In case of accidental overdose, seek professional assistance or contact a poison control center immediately.
All prescription substitutions using this product shall be pursuant to state statutes as applicable. This is not an Orange Book product.
Manufactured by: Sancilio & Company, Inc3874 Fiscal Court #200Riviera Beach, FL 33404 USA
Package Label.principal Display Panel
PRINCIPAL DISPLAY PANEL - 0.25 mg Tablet Label
NDC 44946-1015-3
Sancilio & Company, Inc. Developing Good Science into Great Medicine
LUDENT™Fluoride Chews
Chewable Fluoride Tablets 0.25mg *
*(From 2.2 mg Sodium Fluoride) Sugar and dye free
Rx Only
120 Tablets
Made in the U.S.A.

Package Label.principal Display Panel
PRINCIPAL DISPLAY PANEL - 0.5 mg Tablet Label
NDC 44946-1016-3
Sancilio & Company, Inc. Developing Good Science into Great Medicine
LUDENT™Fluoride Chews
Chewable Fluoride Tablets 0.5mg *
*(From 2.2 mg Sodium Fluoride) Sugar and dye free
Rx Only
120 Tablets
Made in the U.S.A.

Package Label.principal Display Panel
PRINCIPAL DISPLAY PANEL - 1.0 mg Tablet Label
NDC 44946-1017-3
Sancilio & Company, Inc. Developing Good Science into Great Medicine
LUDENT™Fluoride Chews
Chewable Fluoride Tablets 1.0mg *
*(From 2.2 mg Sodium Fluoride) Sugar and dye free
Rx Only
120 Tablets
Made in the U.S.A.

DISCLAIMER:
"This tool does not provide medical advice, and is for informational and educational purposes only, and is not a substitute for professional medical advice, treatment or diagnosis. Call your doctor to receive medical advice. If you think you may have a medical emergency, please dial 911."
"Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. We may limit or otherwise restrict your access to the API in line with our Terms of Service."
"This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product."
PillSync may earn a commission via links on our site