ACETAMINOPHEN (acetaminophen 325 mg) Dailymed
Generic: acetaminophen is used for the treatment of Fever Glucosephosphate Dehydrogenase Deficiency Liver Diseases Pain
Go PRO for all pill images
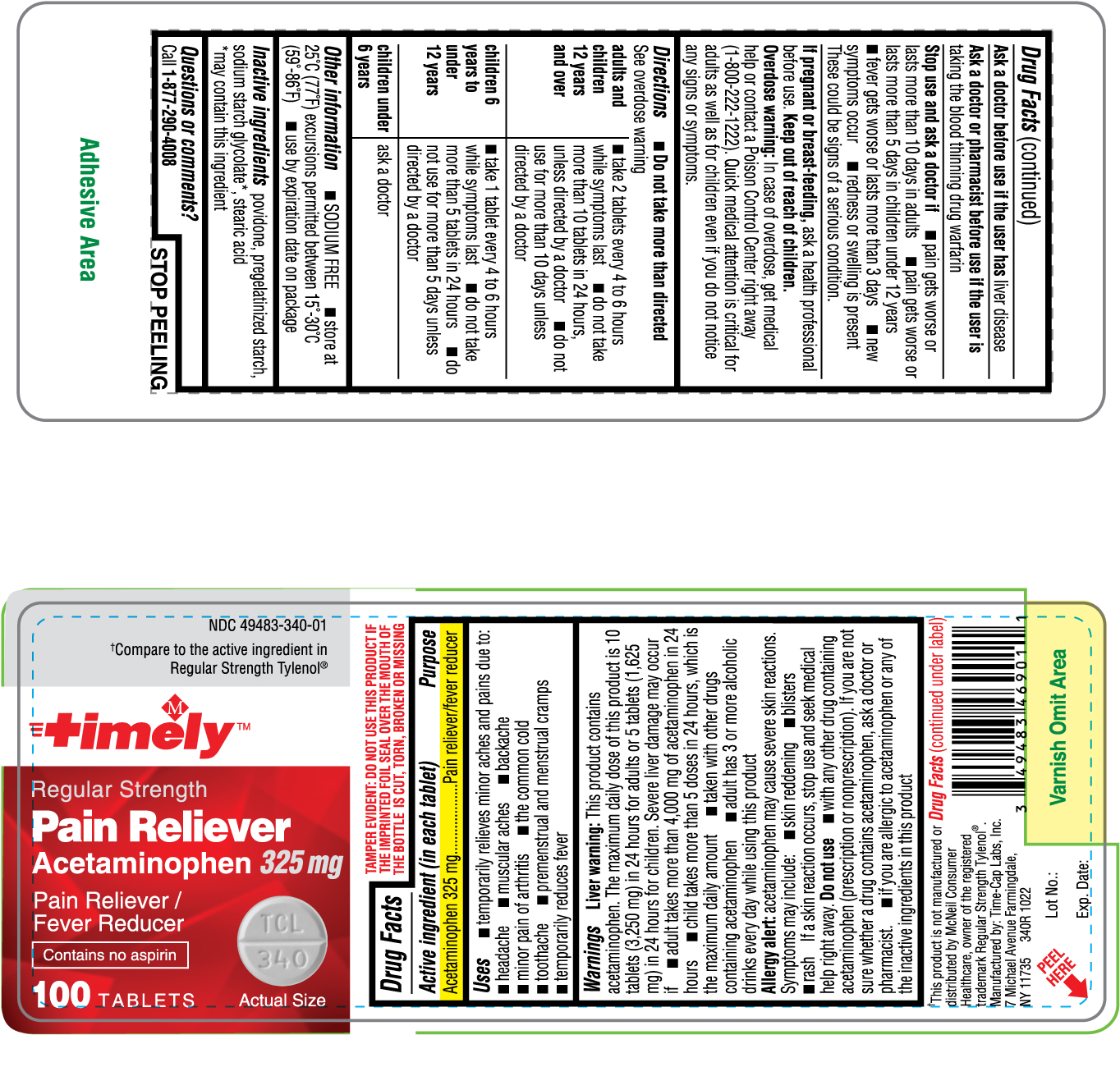
Drug Facts
Otc - Active Ingredient Section
Active ingredient (in each tablet) Acetaminophen 325 mg
Otc - Purpose Section
Purpose Pain reliever/fever reducer
Indications & Usage Section
Uses
- temporarily relieves minor aches and pains due to:
headache
muscular aches
backache
minor pain of arthritis
the common cold
toothache
premenstrual and menstrual cramps
- temporarily reduces fever
Warnings Section
Warnings Liver warning: This product contains acetaminophen. The maximum daily dose of this product is 10 tablets (3,250 mg) in 24 hours for adults or 5 tablets (1,625 mg) in 24 hours for children. Severe liver damage may occur if • adult takes more than 4,000 mg of acetaminophen in 24 hours • child takes more than 5 doses in 24 hours, which is the maximum daily amount • taken with other drugs containing acetaminophen • adult has 3 or more alcoholic drinks every day while using this product Allergy alert: acetaminophen may cause severe skin reactions. Symptoms may include: • skin reddening • bulers • rash
If a skin reaction occurs, stop use and seek medical help right away.
Otc - Do Not Use Section
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are allergic to acetaminophen or any of the inactive ingredients in this product
Otc - Ask Doctor Section
Ask a doctor before use if the user has liver disease
Otc - Ask Doctor/pharmacist Section
Ask a doctor or pharmacist before use if the user is taking the blood thinning drug warfarin
Otc - Stop Use Section
Stop use and ask a doctor if • pain gets worse or lasts more than 10 days in adults • pain gets worse or lasts more than 5 days in children under 12 years • fever gets worse or lasts more than 3 days • new symptoms occur • redness or swelling is present These could be signs of a serious condition.
Otc - Pregnancy Or Breast Feeding Section
If pregnant or breast-feeding, ask a health professional before use.
Otc - Keep Out Of Reach Of Children Section
Keep out of reach of children.
Overdose warning: In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222). Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Dosage & Administration Section
Directions Do not take more than directed. See overdose warning adults and children 12 years and over • take 2 tablets every 4 to 6 hours while symptoms last • do not take more than 10 tablets in 24 hours, unless directed by a doctor • do not use for more than 10 days unless directed by a doctor children 6 years to under 12 years • take 1 tablet every 4 to 6 hours while symptoms last • do not take more than 5 tablets in 24 hours • do not use for more than 5 days unless directed by a doctor children under 6 years • ask a doctor
Other Information
Other information
- SODIUM FREE
- store at 25°C (77°F) excursions permitted between 15°-30°C (59°-86°F)
- use by expiration date on package
Inactive Ingredient Section
Inactive ingredients povidone, pregelatinized starch, sodium starch glycolate*, stearic acid *may contain this ingredient
Package Label.principal Display Panel
DISCLAIMER:
"This tool does not provide medical advice, and is for informational and educational purposes only, and is not a substitute for professional medical advice, treatment or diagnosis. Call your doctor to receive medical advice. If you think you may have a medical emergency, please dial 911."
"Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. We may limit or otherwise restrict your access to the API in line with our Terms of Service."
"This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product."
PillSync may earn a commission via links on our site