BL-Contrast Dailymed
Boxed Warning
Warning: Risks With Inadvertent Intrathecal Administration Omnipaque Injection, 140 And 350 Mg Iodine/ml
Go PRO for all pill images
Warning: Risks With Inadvertent Intrathecal Administration Omnipaque Injection, 140 And 350 Mg Iodine/ml
Inadvertent intrathecal administration may cause death, convulsions/seizures, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, rhabdomyolysis, hyperthermia, and brain edema [see Contraindications (4)and Warnings and Precautions (5.1)].
WARNING: RISKS WITH INADVERTENT INTRATHECAL ADMINISTRATION OF OMNIPAQUE injection 140 and 350 mg iodine/mL
See full prescribing information for complete boxed warning.
Inadvertent intrathecal administration may cause death, convulsions/seizures, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, rhabdomyolysis, hyperthermia, and brain edema (4 ,5.1 ).
Recent Major Changes Section
Warnings and Precautions, Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age ( 5.9 )4/2023
1 Indications And Usage
OMNIPAQUE (iohexol) injection is a radiographic contrast agent indicated for intrathecal, intravascular, oral, rectal, intraarticular and body cavity use. OMNIPAQUE oral solution is indicated for oral use only in conjunction with OMNIPAQUE injection administered intravenously for computed tomography (CT) of the abdomen (1 ).
1.1 Intrathecal Administration
Adults
OMNIPAQUE 180, 240, and 300
- Myelography (lumbar, thoracic, cervical, total columnar)
- Computerized Tomography (CT) (myelography, cisternography, ventriculography)
Pediatrics
OMNIPAQUE 180
- Myelography (lumbar, thoracic, cervical, total columnar)
- CT (myelography, cisternography)
1.2 Intravascular Administration
Adults
OMNIPAQUE 140
- Intra-arterial digital subtraction angiography of the head, neck, abdominal, renal and peripheral vessels
OMNIPAQUE 240
- CT head imaging
- Peripheral venography (phlebography)
OMNIPAQUE 300
- Aortography including studies of the aortic arch, abdominal aorta and its branches
- CT head and body imaging
- Cerebral arteriography
- Peripheral venography (phlebography)
- Peripheral arteriography
- Excretory urography
OMNIPAQUE 350
- Angiocardiography (ventriculography, selective coronary arteriography)
- Aortography including studies of the aortic root, aortic arch, ascending aorta, abdominal aorta and its branches
- CT head and body imaging
- Intravenous digital subtraction angiography of the head, neck, abdominal, renal and peripheral vessels
- Peripheral arteriography
- Excretory urography
Pediatrics
OMNIPAQUE 240
- CT head and body imaging
OMNIPAQUE 300
- Angiocardiography (ventriculography)
- Excretory urography
- CT head and body imaging
OMNIPAQUE 350
- Angiocardiography (ventriculography, pulmonary arteriography, venography, and studies of the collateral arteries)
- Aortography including the aortic root, aortic arch, ascending and descending aorta
1.3 Oral or Rectal Administration
Adults
OMNIPAQUE 350
- Oral radiographic examination of the gastrointestinal tract
Pediatrics
OMNIPAQUE 180, 240 and 300
- Oral and rectal radiographic examination of the gastrointestinal tract
1.4 Oral Administration in Conjunction with Intravenous Administration
Diluted OMNIPAQUE Injection
Adults
OMNIPAQUE 240, 300 and 350 diluted and administered orally in conjunction with OMNIPAQUE 300 administered intravenously
- CT of the abdomen
Pediatrics
OMNIPAQUE 240, 300 and 350 diluted and administered orally in conjunction with OMNIPAQUE 240 or OMNIPAQUE 300 administered intravenously
- CT of the abdomen
OMNIPAQUE Oral Solution
Adults
OMNIPAQUE oral solution 9 and 12 administered orally in conjunction with OMNIPAQUE 300 administered intravenously
- CT of the abdomen
Pediatrics
OMNIPAQUE oral solution 9 and 12 administered orally in conjunction with OMNIPAQUE 240 or OMNIPAQUE 300 administered intravenously
- CT of the abdomen
1.5 Intraarticular Administration
Adults
OMNIPAQUE 240, 300, and 350
- Arthrography
1.6 Body Cavity Administration
Adults
OMNIPAQUE 240
- Endoscopic retrograde pancreatography (ERP) and cholangiopancreatography (ERCP)
- Herniography
- Hysterosalpingography
OMNIPAQUE 300
- Hysterosalpingography
Pediatrics
OMNIPAQUE 240, 300 and 350 diluted
- Voiding cystourethrography (VCU)
2 Dosage And Administration
The concentration and volume required will depend on the indication, size and condition of the patient, and the equipment and imaging technique used. For CT of the head and body, OMNIPAQUE may be used with an automated contrast injection system or contrast media management system cleared for use with OMNIPAQUE. See full prescribing information for complete dosing information (2 ).
2.1 Important Dosage and Administration Instructions
- OMNIPAQUE 140, 180, 240, 300 and 350 are indicated for intravascular, oral, rectal, intraarticular, and body cavity administration. OMNIPAQUE 180, 240, and 300 are indicated for intrathecal administration [see Boxed Warning, Contraindications (4), and Warnings and Precautions (5.1)].
- Use sterile technique for all handling and administration of OMNIPAQUE for intravascular, intrathecal, intraarticular, and body cavity administration.
- OMNIPAQUE oral solution 9 and 12 are indicated for oral use only [see Contraindications (4) and Warnings and Precautions (5.2)].
- Do not use if tamper-evident ring is broken or missing.
- OMNIPAQUE injection may be administered at either body (37°C, 98.6°F) or room temperature (20° to 25°C, 68° to 77°F).
- Inspect OMNIPAQUE injection for particulate matter or discoloration before administration, whenever solution and container permit. Do not administer if OMNIPAQUE injection contains particulate matter or is discolored.
- Do not mix OMNIPAQUE injection with, or inject in intravenous lines containing, other drugs or total nutritional admixtures.
- Use the lowest dose necessary to obtain adequate visualization.
- Individualize the volume, strength, and rate of administration of OMNIPAQUE injection. Consider factors such as age, body weight, vessel size, blood flow rate within the vessel, anticipated pathology, degree and extent of opacification required, structures or area to be examined, disease processes affecting the patient, and equipment and technique to be employed.
- Avoid extravasation when administering OMNIPAQUE injection intravascularly, especially in patients with severe arterial or venous disease [see Warnings and Precautions (5.6)] .
- Hydrate patients before and after intravascular administration of OMNIPAQUE injection [see Warnings and Precautions (5.4) ].
- Each bottle of OMNIPAQUE injection and oral solution is intended for one procedure only. Discard any unused portion.
2.2 Intrathecal Dosage and Administration
- Rate of injection: Injection should be made slowly over 1 to 2 minutes
- Repeat procedures: If sequential or repeat examinations are required, a suitable interval of time between administrations should be observed to allow for normal clearance of the drug from the body; at least 48 hours should be allowed before repeat examination; however, whenever possible, 5 days to 7 days is recommended.
- If computerized tomographic (CT) myelography follows myelography, delay imaging several hours to allow the degree of contrast to decrease.
TABLE 1 - INTRATHECAL ADULTS The usual recommended total doses for use in lumbar, thoracic, cervical, and total columnar myelography in adults are 1,200 mg iodine to 3,100 mg iodine (see below). STUDY TYPE INJECTION TYPE CONCENTRATION (mg iodine/mL) VOLUME (mL) * A total dose of 3,100 mg iodine or a concentration of 300 mg iodine/mL should not be exceeded in adults. LUMBAR MYELOGRAPHY LUMBAR OMNIPAQUE 180 OMNIPAQUE 240 10 to 17 7 to 12.5 THORACIC MYELOGRAPHY LUMBAR CERVICAL OMNIPAQUE 240 OMNIPAQUE 300 6 to 12.5 6 to 10 CERVICAL MYELOGRAPHY LUMBAR OMNIPAQUE 240 OMNIPAQUE 300 6 to 12.5 6 to 10 CERVICAL MYELOGRAPHY C1-2 OMNIPAQUE 180 OMNIPAQUE 240 OMNIPAQUE 300 7 to 10 6 to 12.5 4 to 10 TOTAL COLUMNAR MYELOGRAPHY LUMBAR OMNIPAQUE 240 OMNIPAQUE 300 6 to 12.5 6 to 10
TABLE 2 – INTRATHECAL PEDIATRICS The usual recommended total doses for lumbar, thoracic, cervical, and/or total columnar myelography by lumbar puncture in children are 360 mg iodine to 2700 mg iodine (see below). Actual volumes administered depend largely on patient age and the following guidelines are recommended. AGE STUDY TYPE INJECTION TYPE CONCENTRATION (mg iodine/mL) VOLUME (mL) *A total dose of 2,700 mg iodine or a concentration of 180 mg iodine/mL should not be exceeded in a single myelographic examination in pediatrics. 0 up to 3 mos. LUMBAR, THORACIC, CERVICAL AND/OR TOTAL COLUMNAR MYELOGRAPHY LUMBAR PUNCTURE OMNIPAQUE 180 2 to 4 3 up to 36 mos. OMNIPAQUE 180 4 to 8 3 up to 7 yrs. OMNIPAQUE 180 5 to 10 7 up to 13 yrs. OMNIPAQUE 180 5 to 12 13 to 18 yrs. OMNIPAQUE 180 6 to 15 2.3 Intravascular Dosage and Administration
Intra-arterial Procedures
TABLE 3 ANGIOCARDIOGRAPHIC PROCEDURES PATIENT POPULATION CONCENTRATION (mg iodine/mL) VOLUME (mL) Adults OMNIPAQUE 350 VENTRICULOGRAPHY SELECTIVE CORONARY ARTERIOGRAPHY
- The recommended single dose is 40 mL (Range of 30 mL to 60 mL)
- May be combined with selective coronary arteriography
Doses may be repeated as necessary. Maximum volume with multiple injections should not exceed 250 mL.
- The recommended single dose is 5 mL (Range of 3 mL to 14 mL)
Pediatrics OMNIPAQUE 300 VENTRICULOGRAPHY The recommended single dose is 1.75 mL/kg (Range of 1.5 mL/kg to 2 mL/kg) Maximum dose with multiple injections should not exceed 6 mL/kg up to a total volume of 291 mL.
- May be repeated as necessary
OMNIPAQUE 350 VENTRICULOGRAPHY Recommended single dose is 1.25 mL/kg (Range of 1 mL/kg to 1.5 mL/kg). Maximum dose with multiple injections should not exceed 5 mL/kg up to a total volume of 250 mL. PULMONARY ANGIOGRAPHY (PULMONARY ARTERIOGRAPHY AND/OR PULMONARY VENOGRAPHY) The recommended single dose is 1 mL/kg.
- May be repeated as necessary
TABLE 4 AORTOGRAPHY PATIENT POPULATION CONCENTRATION (mg iodine/mL) VOLUME (mL) Adults OMNIPAQUE 300 and 350 AORTOGRAPHY AND SELECTIVE VISCERAL ARTERIOGRAPHY The recommended single dose is: Injections may be repeated if indicated, but the total volume should not exceed:
- 50 mL to 80 mL for the aorta (aortic arch, ascending aorta)
- 30 mL to 60 mL for abdominal aorta and its branches (celiac, mesenteric, hepatic and splenic arteries)
- 5 mL to 15 mL for renal arteries
- 290 mL of OMNIPAQUE 300
- 250 mL of OMNIPAQUE 350
OMNIPAQUE 350 AORTIC ROOT AND ARCH STUDY WHEN USED ALONE The recommended single dose is 50 mL (Range of 20 mL to 75 mL) Pediatrics OMNIPAQUE 350 AORTOGRAPHY (AORTIC ROOT, AORTIC ARCH, AND DESCENDING AORTA) The recommended single dose is 1 mL/kg. Maximum dose should not exceed 5 mL/kg up to a total volume of 250 mL.
- May be repeated as necessary
TABLE 5 CEREBRAL ARTERIOGRAPHY PATIENT POPULATION CONCENTRATION (mg iodine/mL) VOLUME (mL) Adults OMNIPAQUE 300 Single dose for cerebral arteriography is as follows:
- Common carotid artery (6 mL to 12 mL)
- Internal carotid artery (8 mL to 10 mL)
- External carotid artery (6 mL to 9 mL)
- Vertebral artery (6 mL to 10 mL)
TABLE 6 INTRA-ARTERIAL DIGITAL SUBTRACTION ANGIOGRAPHY HEAD, NECK, ABDOMINAL, RENAL AND PERIPHERAL VESSELS PATIENT POPULATION CONCENTRATION (mg iodine/mL) VOLUME (mL) Mechanical or hand injection can be used to administer one or more bolus intra-arterial injections of OMNIPAQUE 140. Adults OMNIPAQUE 140 ARTERIES VOLUME/INJECTION (mL) RATE OF INJECTION (mL/sec) Aorta 20 to 45 8 to 20 Carotid 5 to 10 3 to 6 Femoral 9 to 20 3 to 6 Vertebral 4 to 10 2 to 8 Renal 6 to 12 3 to 6 Other branches of aorta (includes subclavian, axillary, innominate and iliac) 8 to 25 3 to 10
TABLE 7 PERIPHERAL ARTERIOGRAPHY PATIENT POPULATION CONCENTRATION (mg iodine/mL) VOLUME (mL) Adults OMNIPAQUE 300 and 350 The recommended dose for use in peripheral angiography is as follows: Aortofemoral runoffs: Selective arteriograms:
- 30 mL to 90 mL of OMNIPAQUE 300
- 20 mL to 70 mL of OMNIPAQUE 350
- 10 mL to 60 mL of OMNIPAQUE 300
- 10 mL to 30 mL of OMNIPAQUE 350
Intravenous Procedures
TABLE 8 PERIPHERAL VENOGRAPHY (PHLEBOGRAPHY) PATIENT POPULATION CONCENTRATION (mg iodine/mL) VOLUME (mL) Adults OMNIPAQUE 240 and 300 The recommended dose (per leg) is:
- 20 mL to 150 mL of OMNIPAQUE 240
- 40 mL to 100 mL of OMNIPAQUE 300
TABLE 9 EXCRETORY UROGRAPHY PATIENT POPULATION CONCENTRATION (mg iodine/mL) VOLUME (mL) Adults OMNIPAQUE 300 and 350 The recommended dose is:
- 0.6 mL/kg to 1.2 mL/kg body weight
Pediatrics OMNIPAQUE 300 Dose ranging from 0.5 mL/kg to 3 mL/kg of body weight:
- The usual dose for children is 1 mL/kg to 1.5 mL/kg.
- The total administered dose should not exceed 3 mL/kg.
TABLE 10 DIGITAL SUBTRACTION ANGIOGRAPHY HEAD, NECK, ABDOMINAL, RENAL AND PERIPHERAL VESSELS PATIENT POPULATION CONCENTRATION (mg iodine/mL) VOLUME (mL) RATE OF INJECTION (mL/sec) Adults OMNIPAQUE 350 The usual dose for the intravenous digital technique is 30 mL to 50 mL. Frequently three or more doses may be required, up to a total volume not to exceed 250 mL 7.5 mL/second to 30 mL/second using a pressure injector
TABLE 11 CT SCANNING OF THE HEAD AND BODY PATIENT POPULATION CONCENTRATION (mg iodine/mL) VOLUME OMNIPAQUE may be used with an automated contrast injection system or contrast management system cleared for use with OMNIPAQUE [see Dosage and Administration (2.8)]. See device labeling for device indications, additional information, and instructions for use. (mL)Adults OMNIPAQUE 240, 300 and 350 Head and body imaging by rapid injection CT Imaging – Head: CT Imaging – Body:
- 70 mL to 150 mL of OMNIPAQUE 300
- 80 mL of OMNIPAQUE 350
Head imaging by infusion CT Imaging – Head:
- 50 mL to 200 mL of OMNIPAQUE 300
- 60 mL to 100 mL of OMNIPAQUE 350
- 120 mL to 250 mL of OMNIPAQUE 240
Pediatrics OMNIPAQUE 240 and 300 CT Imaging – Head and Body:
- 1 mL/kg to 2 mL/kg (with maximum = 3 mL/kg)
- Maximum single dose = 116 mL
2.4 Oral or Rectal Dosage and Administration
Oral and Rectal Administration – Undiluted OMNIPAQUE Injection for Radiographic Examination of the Gastrointestinal (GI) Tract
TABLE 12 DOSING FOR RADIOGRAPHIC EXAMINATION OF THE GI TRACT PATIENT POPULATION CONCENTRATION (mg iodine/mL) ORAL VOLUME (mL) RECTAL VOLUME When given rectally, larger volumes may be used. (mL)Adults OMNIPAQUE 350 The recommended dose is 50 mL to 100 mL - Pediatrics OMNIPAQUE 180, 240 and 300 The recommended dose is 5 mL to 100 mL The recommended dose is 5 mL to 100 mL Less than 3 months old OMNIPAQUE 180 5 mL to 30 mL - Three months to 3 years OMNIPAQUE 180, 240 and 300 Up to 60 mL - Four years to 10 years OMNIPAQUE 180, 240 and 300 Up to 80 mL - Greater than 10 years Up to 100 mL - 2.5 Oral Dosage and Administration in Conjunction with Intravenous Administration
See Table 16for concurrent intravenous dosing.
Oral Administration of Diluted OMNIPAQUE Injection in Conjunction with Intravenous Administration of OMNIPAQUE Injection for CT of the Abdomen
TABLE 13 DOSING OF DILUTED Dilutions of OMNIPAQUE should be prepared just prior to use and any unused portion discarded after the procedure. OMNIPAQUE INJECTION FOR ORAL ADMINISTRATIONPATIENT POPULATION ORAL CONCENTRATION (mg iodine/mL) ORAL VOLUME (mL) ADMINISTRATION INSTRUCTIONS Adults OMNIPAQUE 240, 300 and 350 DILUTED to 6 to 12 mg iodine/mL (See Table 14below) Recommended oral dose is:
- 500 mL to 1,000 mL
Smaller administered volumes can be given if the iodine concentration in final diluted product is increased (See Table 14below) The oral dosage may be given all at once or over a period of up to 45 minutes if there is difficulty in consuming the required volume. Pediatrics OMNIPAQUE 240, 300 and 350 DILUTED to 9 to 21 mg iodine/mL (See Table 14below) Recommended oral dose is: Do not exceed an oral dose of 5 grams iodine for patients less than 3 years old. Do not exceed an oral dose of 10 grams iodine for patients 3 to 18 years old.
- 180 mL to 750 mL
Smaller administered volumes can be given if the iodine concentration in final diluted product is increased (See Table 14below) The oral dosage may be given all at once or over a period of up to 45 minutes if there is difficulty in consuming the required volume.
TABLE 14 PROCEDURE FOR PREPARATION OF DILUTED OMNIPAQUE INJECTION FOR ORAL ADMINISTRATION OMNIPAQUE to be mixed with liquid such as water, carbonated beverage, milk, infant formula, or juice to achieve one liter of oral contrast agent. Final Iodine Concentration of Diluted Contrast Agent (mg iodine/mL) OMNIPAQUE 240 OMNIPAQUE 300 OMNIPAQUE 350 Volume of Contrast Agent (mL) Volume of Liquid (mL) Volume of Contrast Agent (mL) Volume of Liquid (mL) Volume of Contrast Agent (mL) Volume of Liquid (mL) 6 25 975 20 980 17 983 9 38 962 30 970 26 974 12 50 950 40 960 35 965 15 63 937 50 950 43 957 18 75 925 60 940 52 948 21 88 912 70 930 60 940
Oral Administration of OMNIPAQUE Oral Solution in Conjunction with Intravenous Administration of OMNIPAQUE Injection for CT of the Abdomen
TABLE 15 DOSING AND ADMINISTRATION OF OMNIPAQUE ORAL SOLUTION PATIENT POPULATION ORAL CONCENTRATION (mg iodine/mL) ORAL VOLUME (mL) ADMINISTRATION INSTRUCTIONS Adults OMNIPAQUE oral solution 9 and 12 The recommended oral dose is:
- 500 mL to 1,000 mL
The oral dosage may be given all at once or over a period of up to 45 minutes if there is difficulty in consuming the required volume. Pediatrics OMNIPAQUE oral solution 9 and 12 The recommended oral dose is: Do not exceed an oral dose of 5 grams iodine for patients less than 3 years old. Do not exceed an oral dose of 10 grams iodine for patients 3 to 18 years old.
- 180 mL to 750 mL
The oral dosage may be given all at once or over a period of up to 45 minutes if there is difficulty in consuming the required volume.
TABLE 16 INTRAVENOUS ADMINISTRATION OF OMNIPAQUE INJECTION FOR CT OF THE ABDOMEN IN CONJUNCTION WITH ORALLY ADMINISTERED DILUTED OMNIPAQUE INJECTION OR OMNIPAQUE ORAL SOLUTION PATIENT POPULATION INTRAVENOUS CONCENTRATION (mg iodine/mL) INTRAVENOUS VOLUME OMNIPAQUE may be used with an automated contrast injection system or contrast management system cleared for use with OMNIPAQUE [see Dosage and Administration (2.8)]. See device labeling for device indications, additional information, and instructions for use. (mL)ADMINISTRATION INSTRUCTIONS Adults OMNIPAQUE 300 The recommended dose is:
- 100 mL to 150 mL
Administer up to 40 minutes AFTER consumption of the oral dose Pediatrics OMNIPAQUE 240 and 300 The recommended dose is:
- 2 mL/kg with a range of 1 mL/kg to 2 mL/kg (maximum 3 mL/kg)
Administer up to 60 minutes AFTER consumption of the oral dose 2.6 Intraarticular Dosage and Administration
TABLE 17 ARTHROGRAPHY PATIENT POPULATION LOCATION CONCENTRATION (mg iodine/mL) VOLUME (mL) DOUBLE CONTRAST/SINGLE CONTRAST Adults Knee Passive or active manipulation is used to disperse the medium throughout the joint space. OMNIPAQUE 240 5 to 15 Lower volumes recommended for double-contrast examinations; higher volumes recommended for single-contrast examinations. OMNIPAQUE 300 5 to 15 OMNIPAQUE 350 5 to 10 Adults Shoulder OMNIPAQUE 240 3 OMNIPAQUE 300 10 Adults Temporomandibular OMNIPAQUE 300 0.5 to 1 2.7 Body Cavity Dosage and Administration
Body Cavity Administration - Undiluted OMNIPAQUE Injection
TABLE 18 ENDOSCOPIC RETROGRADE PANCREATOGRAPHY (ERP) ENDOSCOPIC RETROGRADE CHOLANGIOPANCREATOGRAPHY (ECRP) PATIENT POPULATION CONCENTRATION (mg iodine/mL) VOLUME (mL) Adults OMNIPAQUE 240 10 mL to 50 mL but may vary depending on individual anatomy and/or disease state.
TABLE 19 HYSTEROSALPINGOGRAPHY PATIENT POPULATION CONCENTRATION (mg iodine/mL) VOLUME (mL) Adults OMNIPAQUE 240 and 300 15 mL to 20 mL but may vary depending on individual anatomy and/or disease state.
TABLE 20 HERNIOGRAPHY PATIENT POPULATION CONCENTRATION (mg iodine/mL) VOLUME (mL) Adults OMNIPAQUE 240 50 mL but may vary depending on individual anatomy and/or disease state.
Body Cavity Administration - Diluted OMNIPAQUE Injection
TABLE 21 VOIDING CYSTOURETHROGRAPHY (VCU) (CAN BE PERFORMED IN CONJUNCTION WITH EXCRETORY UROGRAPHY) PATIENT POPULATION CONCENTRATION (mg iodine/mL) VOLUME (mL) Pediatrics The concentration may vary depending upon the patient's size and age and with the technique and equipment used. OMNIPAQUE injection may be diluted with Sterile Water for Injection. (See Table 22below). OMNIPAQUE injection may be diluted, utilizing aseptic technique, with Sterile Water for Injection to a concentration of 50 mg iodine/mL to 100 mg iodine/mL for voiding cystourethrography. Range:
- 50 mL to 300 mL of DILUTED OMNIPAQUE at a concentration of 100 mg iodine/mL
- 50 mL to 600 mL of DILUTED OMNIPAQUE at a concentration of 50 mg iodine/mL.
TABLE 22 PROCEDURE FOR PREPARATION OF DILUTED Dilutions of OMNIPAQUE should be prepared just prior to use and any unused portion discarded after the procedure. OMNIPAQUE INJECTION FOR VCUFinal Iodine Concentration of Diluted Contrast Agent (mg iodine/mL) Volume of OMNIPAQUE 240 (mL) Volume of Sterile Water for Injection (mL) Volume of OMNIPAQUE 300 (mL) Volume of Sterile Water for Injection (mL) Volume of OMNIPAQUE 350 (mL) Volume of Sterile Water for Injection (mL) 100 100 140 100 200 100 250 90 167 233 289 80 200 275 338 70 243 330 400 60 300 400 483 50 380 500 600 2.8 Instructions for Use with an Automated Contrast Injection System or Contrast Management System for CT of the Head and Body
- OMNIPAQUE may be used with an automated contrast injection system cleared for use with contrast media.
- See above Important Dosage and Administration Instructions for OMNIPAQUE (2.1).
- See device labeling for information on device indications, instructions for use, and techniques to help assure safe use.
- OMNIPAQUE 300 mg iodine/mL and 350 mg iodine/mL in 150 mL bottles may be used with a contrast media management system cleared for use with OMNIPAQUE 300 mg iodine/mL and 350 mg iodine/mL in 150 mL bottles.
- See device labeling for information on device indications, instructions for use, and techniques to help assure safe use.
- Use sterile technique for penetrating the container closure of OMNIPAQUE 300 and 350 and transferring OMNIPAQUE solution. The container closure may be penetrated only one time with a suitable sterile component of the contrast media management system cleared for use with OMNIPAQUE 300 and 350 in 150 mL bottles.
- Once the OMNIPAQUE 300 and 350 Injection is punctured, do not remove the bottle from the work area during the entire period of use.
- Maximum use time is 4 hours after initial puncture.
- Each bottle is for one procedure only. Discard unused portion.
3 Dosage Forms And Strengths
OMNIPAQUE Injection
- 140 mg of iodine per mL (302 mg of iohexol/mL) in +PlusPak™ polymer bottles
- 180 mg of iodine per mL (388 mg of iohexol/mL) in glass vials
- 240 mg of iodine per mL (518 mg of iohexol/mL), 300 mg of iodine per mL (647 mg of iohexol/mL) and 350 mg of iodine per mL (755 mg of iohexol/mL) in glass vials and bottles and +PlusPak™ polymer bottles
OMNIPAQUE Oral Solution
- 9 mg of iodine per mL (19 mg of iohexol/mL) and 12 mg of iodine per mL (26 mg of iohexol/mL) in +PlusPak™ polymer bottles
OMNIPAQUE (iohexol) Injection and Oral Solution
Sterile, pyrogen-free, gluten-free, colorless to pale yellow solution containing the nonionic, water-soluble x-ray contrast medium iohexol, and available in the following strengths and formats:
OMNIPAQUE (iohexol) Injection
- 140 mg of organically bound iodine per mL (302 mg iohexol/mL)
- Available in + PLUSPAK™ (polymer bottle)
- 180 mg of organically bound iodine per mL (388 mg iohexol/mL)
- Available in glass vials
- 240 mg of organically bound iodine per mL (518 mg iohexol/mL)
- 300 mg of organically bound iodine per mL (647 mg iohexol/mL)
- 350 mg of organically bound iodine per mL (755 mg iohexol/mL)
- Available in glass vials and bottles and + PLUSPAK™ polymer bottles.
OMNIPAQUE Oral Solution
- 9 mg of organically bound iodine per mL (19 mg iohexol/mL)
- 12 mg of organically bound iodine per mL (26 mg iohexol/mL)
- Available in + PLUSPAK™ polymer bottles.
4 Contraindications
- OMNIPAQUE 140 and OMNIPAQUE 350 are contraindicated for intrathecal use [ see Warnings and Precautions (5.1)]
- OMNIPAQUE oral solution 9 and 12 are contraindicated for parenteral administration [ see Warnings and Precautions (5.2)]
- OMNIPAQUE body cavity 240 and 300 for hysterosalpingography is contraindicated during pregnancy or suspected pregnancy, menstruation or when menstruation is imminent, within 6 months after termination of pregnancy, within 30 days after conization or curettage, when signs of infection are present in any portion of the genital tract including the external genitalia, and when reproductive tract neoplasia is known or suspected because of the risk of peritoneal spread of neoplasm.
- OMNIPAQUE injection 140 and 350 are contraindicated for intrathecal use (
4 )- OMNIPAQUE oral solution 9 and 12 are contraindicated for parenteral use (
4 )- OMNIPAQUE body cavity 240 and 300 for hysterosalpingography is contraindicated during pregnancy (or suspected pregnancy), menstruation (or when menstruation is imminent), within 6 months after termination of pregnancy, within 30 days after conization or curettage, when signs of infection are present in any portion of the genital tract, including the external genitalia, and when reproductive tract neoplasia is known or suspected. (
4 )
5 Warnings And Precautions
- Hypersensitivity Reactions: Life-threatening or fatal reactions can occur. Always have emergency equipment and trained personnel available. (
5.3 )- Contrast-Induced Acute Kidney Injury: Acute injury including renal failure can occur. Minimize dose and maintain adequate hydration to minimize risk. (
5.4 )- Cardiovascular Adverse Reactions: Hemodynamic disturbances including shock and cardiac arrest may occur during or after administration. (
5.5 )- Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age: Individualize thyroid function monitoring based on risk factors such as prematurity. (
5.9 )5.1 Risks Associated with Inadvertent Intrathecal Administration
OMNIPAQUE injection 140 and 350 are contraindicated for intrathecal use [see Contraindications (4)and Dosage and Administration (2.1)]. Inadvertent intrathecal administration can cause death, convulsions/seizures, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, rhabdomyolysis, hyperthermia, and brain edema.
5.2 Risks Associated with Inadvertent Parenteral Administration
OMNIPAQUE oral solution 9 and 12 are contraindicated for parenteral administration [ see Contraindications (4)and Dosage and Administration (2.1)]. Adverse reactions such as hemolysis may occur if administered intravascularly. Do not administer OMNIPAQUE oral solution 9 and 12 parenterally.
5.3 Hypersensitivity Reactions
OMNIPAQUE can cause life-threatening or fatal hypersensitivity reactions including anaphylaxis. Manifestations include respiratory arrest, laryngospasm, bronchospasm, angioedema, and shock. Most severe reactions develop shortly after the start of the injection (within 3 minutes), but reactions can occur up to hours later. There is an increased risk in patients with a history of a previous reaction to contrast agent, and known allergies (i.e., bronchial asthma, drug, or food allergies) or other hypersensitivities. Premedication with antihistamines or corticosteroids does not prevent serious life-threatening reactions, but may reduce both their incidence and severity.
Obtain a history of allergy, hypersensitivity, or hypersensitivity reactions to iodinated contrast agents and always have emergency resuscitation equipment and trained personnel available prior to OMNIPAQUE administration. Monitor all patients for hypersensitivity reactions.
5.4 Contrast-Induced Acute Kidney Injury
Acute kidney injury, including renal failure, may occur after parenteral administration of OMNIPAQUE. Risk factors include: pre-existing renal impairment, dehydration, diabetes mellitus, congestive heart failure, advanced vascular disease, elderly age, concomitant use of nephrotoxic or diuretic medications, multiple myeloma/paraproteinaceous diseases, repetitive and/or large doses of an iodinated contrast agent.
Use the lowest necessary dose of OMNIPAQUE in patients with renal impairment. Adequately hydrate patients prior to and following parenteral administration of OMNIPAQUE. Do not use laxatives, diuretics, or preparatory dehydration prior to OMNIPAQUE administration.
5.5 Cardiovascular Adverse Reactions
Life-threatening or fatal cardiovascular reactions including hypotension, shock, cardiac arrest have occurred with the parenteral administration of OMNIPAQUE. Most deaths occur during injection or five to ten minutes later, with cardiovascular disease as the main aggravating factor. Cardiac decompensation, serious arrhythmias, and myocardial ischemia or infarction can occur during coronary arteriography and ventriculography.
Based upon clinical literature reported deaths from the administration of iodinated contrast agents range from 6.6 per million (0.00066%) to 1 in 10,000 (0.01%). Use the lowest necessary dose of OMNIPAQUE in patients with congestive heart failure and always have emergency resuscitation equipment and trained personnel available. Monitor all patients for severe cardiovascular reactions.
5.6 Thromboembolic Events
Angiocardiography
Serious, rarely fatal, thromboembolic events causing myocardial infarction and stroke can occur during angiocardiography procedures with both ionic and nonionic contrast media. During these procedures, increased thrombosis and activation of the complement system occurs. Risk factors for thromboembolic events include: length of procedure, catheter and syringe material, underlying disease state, and concomitant medications.
To minimize thromboembolic events, use meticulous angiographic techniques, and minimize the length of the procedure. Avoid blood remaining in contact with syringes containing iodinated contrast agents, which increases the risk of clotting. Avoid angiocardiography in patients with homocystinuria because of the risk of inducing thrombosis and embolism.
5.7 Extravasation and Injection Site Reactions
Extravasation of OMNIPAQUE during intravascular injection may cause tissue necrosis and/or compartment syndrome, particularly in patients with severe arterial or venous disease. Ensure intravascular placement of catheters prior to injection. Monitor patients for extravasation and advise patients to seek medical care for progression of symptoms.
5.8 Thyroid Storm in Patients with Hyperthyroidism
Thyroid storm has occurred after the intravascular use of iodinated contrast agents in patients with hyperthyroidism, or with an autonomously functioning thyroid nodule. Evaluate the risk in such patients before use of OMNIPAQUE.
5.9 Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age
Thyroid dysfunction characterized by hypothyroidism or transient thyroid suppression has been reported after both single exposure and multiple exposures to iodinated contrast media (ICM) in pediatric patients 0 to 3 years of age.
Younger age, very low birth weight, prematurity, underlying medical conditions affecting thyroid function, admission to neonatal or pediatric intensive care units, and congenital cardiac conditions are associated with an increased risk of hypothyroidism after ICM exposure. Pediatric patients with congenital cardiac conditions may be at the greatest risk given that they often require high doses of contrast during invasive cardiac procedures.
An underactive thyroid during early life may be harmful for cognitive and neurological development and may require thyroid hormone replacement therapy. After exposure to ICM, individualize thyroid function monitoring based on underlying risk factors, especially in term and preterm neonates.
5.10 Hypertensive Crisis in Patients with Pheochromocytoma
Hypertensive crisis has occurred after the use of iodinated contrast agents in patient with pheochromocytoma. Monitor patients when administering OMNIPAQUE intravascularly if pheochromocytoma or catecholamine-secreting paragangliomas are suspected. Inject the minimum amount of contrast necessary, assess the blood pressure throughout the procedure, and have measures for treatment of a hypertensive crisis readily available.
5.11 Sickle Cell Crisis in Patients with Sickle Cell Disease
Iodinated contrast agents when administered intravascularly may promote sickling in individuals who are homozygous for sickle cell disease. Hydrate patients prior to and following OMNIPAQUE administration and use OMNIPAQUE only if the necessary imaging information cannot be obtained with alternative imaging modalities.
5.12 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions (SCAR) may develop from 1 hour to several weeks after intravascular contrast agent administration. These reactions include Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), acute generalized exanthematous pustulosis (AGEP) and drug reaction with eosinophilia and systemic symptoms (DRESS). Reaction severity may increase and time to onset may decrease with repeat administration of contrast agents; prophylactic medications may not prevent or mitigate severe cutaneous adverse reactions. Avoid administering OMNIPAQUE to patients with a history of a severe cutaneous adverse reaction to OMNIPAQUE.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Risks Associated with Inadvertent Intrathecal Administration [see Warnings and Precautions (5.1)]
- Risks Associated with Inadvertent Parenteral Administration [see Warnings and Precautions (5.2)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.3)]
- Contrast-Induced Kidney Injury [see Warnings and Precautions (5.4)]
- Cardiovascular Adverse Reactions [see Warnings and Precautions (5.5)]
- Thromboembolic Events [see Warnings and Precautions (5.6) ]
- Thyroid Dysfunction in Pediatric Patients 0 to 3 Years of Age [see Warnings and Precautions (5.9)]
- Severe Cutaneous Adverse Reactions [ see Warnings and Precautions (5.12)]
Most common adverse reactions (incidence ≥ 1.0%) in adult patients after OMNIPAQUE administration.(6.1 )
- Intrathecal: Headaches, Pain including backache, neckache, stiffness and neuralgia, nausea, vomiting and dizziness
- Intravascular: Pain, vision abnormalities (including blurred vision and photomas), headache, taste perversion, arrhythmias including premature ventricular contractions (PVCs) and premature atrial contractions (PACs), angina/chest pain, nausea
- Oral: Diarrhea, nausea, vomiting, abdominal pain, flatulence, headache
- Body Cavity: Pain, swelling and heat sensation
Post-marketing adverse reactions (6.2 ):Hypersensitivity and manifestations like rash, pruritus, urticaria, and dyspnea, in addition chest pain, and swelling.
To report SUSPECTED ADVERSE REACTIONS, contact GE Healthcare at 1-800-654-0118 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Intrathecal Administration
Adults
TABLE 23 ADVERSE REACTIONS – INTRATHECAL ADMINISTRATION In controlled clinical studies involving 1531 patients using OMNIPAQUE the following adverse reactions were reported: System Organ Class Adverse Reaction Incidence Nervous System Headaches 18% Musculoskeletal and Connective Tissue Pain including backache, neckache, stiffness and neuralgia 8% Gastrointestinal System Nausea 6% Vomiting 3% Nervous System Dizziness 2% Other Reactions Feeling of heaviness, hypotension, hypertonia, sensation of heat, sweating, vertigo, loss of appetite, drowsiness, hypertension, photophobia, tinnitus, neuralgia, paresthesia, difficulty in micturition, and neurological changes <0.1%
Pediatric Patients
TABLE 24 ADVERSE REACTIONS – INTRATHECAL ADMINISTRATION In clinical studies involving 152 patients for pediatric myelography by lumbar puncture, adverse events following the use of OMNIPAQUE 180 were generally similar to those reported in adults. Procedure System Organ Class Adverse Reaction Incidence Myelography by Lumbar Puncture Nervous System Headache 9% Gastrointestinal System Vomiting 6% Musculoskeletal and Connective Tissue Backache 1.3% Other Reactions All were transient and mild with no clinical sequelae. Fever <0.7% Hives Stomachache Visual Hallucination Neurological Changes
Intravascular Administration
Immediately following intravascular injection of contrast medium, a transient sensation of mild warmth is not unusual. Warmth is less frequent with OMNIPAQUE than with ionic contrast media.
Adults
In controlled clinical studies involving 1485 patients, the following adverse reactions occurred (Table 25).
TABLE 25 ADVERSE REACTIONS – INTRAVASCULAR ADMINISTRATION System Organ Class Adverse Reaction Incidence Cardiovascular System Arrhythmias including PVCs and PACs 2% Hypotension 0.7% Others including cardiac failure, asystole, bradycardia, tachycardia, and vasovagal reaction ≤ 0.3% Nervous System Vertigo (including dizziness and lightheadedness) 0.5% Pain 3% Vision Abnormalities (including blurred vision and photomas) 2% Taste Perversion 1% Other Reactions Anxiety, fever, motor and speech dysfunction, convulsion, paresthesia, somnolence, stiff neck, hemiparesis, syncope, shivering, transient ischemic attack, cerebral infarction, and nystagmus Individual incidence of 0.3% or less Respiratory System Dyspnea, rhinitis, coughing, and laryngitis Individual incidence of 0.2% or less Gastrointestinal System Nausea 2% Vomiting 0.7% Others including diarrhea, dyspepsia, cramp, and dry mouth Individual incidence of less than 0.1%. Skin and Subcutaneous Tissues Urticaria 0.3% Purpura 0.1% Abscess 0.1% Pruritus 0.1%
Pediatric Patients
In controlled clinical studies involving 391 patients for pediatric angiocardiography, urography, and CT head imaging, adverse reactions following the use of OMNIPAQUE 240, 300, and 350 were generally similar in quality and frequency to those reported in adults (Table 26).
TABLE 26 ADVERSE REACTIONS – INTRAVASCULAR ADMINISTRATION System Organ Class Adverse Reaction Incidence Cardiovascular System Ventricular Tachycardia 0.5% 2:1 Heart Block 0.5% Hypertension 0.3% Anemia 0.3% General Disorders and Administration Site Conditions Pain 0.8% Fever 0.5% Nervous System Convulsion 0.3% Taste Abnormality 0.5% Respiratory System Congestion 0.3% Apnea 0.3% Gastrointestinal System Nausea 1% Vomiting 2% Endocrine System Hypoglycemia 0.3% Skin and Subcutaneous Tissue Rash 0.3%
Oral Administration for Examination of the Gastrointestinal Tract
Adults
Nausea, vomiting, and diarrhea have been most frequently reported following orally administered undiluted OMNIPAQUE for radiographic examination of the gastrointestinal tract. In controlled clinical studies involving 54 adult patients for oral radiographic examination of the gastrointestinal tract using undiluted OMNIPAQUE 350 the following adverse reactions were reported (Table 27).
TABLE 27 ADVERSE REACTIONS – ORAL ADMINISTRATION OF UNDILUTED OMNIPAQUE 350 System Organ Class Adverse Reaction Incidence Gastrointestinal System Diarrhea 42% Nausea 15% Vomiting 11% Abdominal Pain 7% Flatulence 2% Nervous System Headache 2%
Pediatrics Patients (Oral and Rectal Administration)
In clinical studies involving 58 pediatric patients, the adverse reactions were found to mostly affect the gastrointestinal system with diarrhea (36%), vomiting (9%), nausea (5%) and abdominal pain (2%). However, fever (5%), hypotension (2%) and urticaria (2%) were also reported.
Oral Administration for CT of the Abdomen in Conjunction with Intravenous Administration
Adults
In a controlled clinical study involving 44 adult patients receiving oral administration of diluted OMNIPAQUE (4-9 mg iodine/mL) in conjunction with intravenously injected OMNIPAQUE 300 for CT examination of the abdomen, adverse reactions were limited to a single report of vomiting.
Pediatric Patients
In clinical studies involving 69 pediatric patients receiving oral administration of diluted OMNIPAQUE (9-29 mg iodine/mL) in conjunction with intravenously administered OMNIPAQUE 240 and OMNIPAQUE 300 for CT examination of the abdomen, adverse reactions were limited to a single report of vomiting (1.4%).
Body Cavity Use
Adults
Arthrography : In controlled clinical studies involving 285 adult patients for various body cavity examinations using OMNIPAQUE 240, 300 and 350, the most frequent adverse reactions were administration site reactions: pain 26% and swelling 22%, were exclusively reported for arthrography and were generally related to the procedure rather than the contrast medium. Patients also experienced heat (7%). All other adverse reaction occurred at a rate less than or equal to 1%.
Pediatric Patients
No adverse reactions associated with the use of OMNIPAQUE for VCU procedures were reported in 51 pediatric patients studied.
6.2 Post-marketing Experience
The following additional reactions uled by indication have been identified during post-approval use of OMNIPAQUE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
General
Immune System Disorders:Hypersensitivity reactions, anaphylactic or anaphylactoid reactions, anaphylactic or anaphylactoid shock including life-threatening or fatal anaphylaxis
General Disorders and Administration Site Conditions:Pyrexia, chills, pain and discomfort, asthenia, administration site conditions including extravasation
Intrathecal Administration
Nervous System Disorders:Meningism, aseptic meningitis, seizures or status epilepticus, disorientation, coma, depressed or loss of consciousness, transient contrast-induced toxic encephalopathy (including amnesia, hallucination, paralysis, paresis, speech disorder, aphasia, dysarthria), restlessness, tremors, hypoesthesia
Musculoskeletal and Connective Tissue Disorders:Pain, muscle spasms or spasticity
Psychiatric Disorders:Confusional state, agitation, anxiety
Eye Disorders:Transient visual impairment including cortical blindness
Renal Reactions:Acute kidney injury
Intravascular Administration
Cardiovascular Disorders:Severe cardiac complications (including cardiac arrest, cardiopulmonary arrest), shock, peripheral vasodilatation, palpitations, vasospasm including spasm of coronary arteries, myocardial infarction, syncope, cyanosis, pallor, flushing, chest pain
Hemodynamic Reactions:Vasospasm and thrombophlebitis following intravenous injection
Blood and Lymphatic System Disorders:Neutropenia
Nervous System Disorders:Disorientation, coma, depressed or loss of consciousness, transient contrast-induced toxic encephalopathy (including amnesia, hallucination, paralysis, paresis, speech disorder, aphasia, dysarthria), restlessness, tremors, hypoesthesia
Psychiatric Disorders:Confusional state, agitation
Eye Disorders:Eye irritation or itchiness, periorbital edema, ocular or conjunctival hyperemia, lacrimation
Renal Reactions:Acute kidney injury, toxic nephropathy (CIN), transient proteinuria, oliguria or anuria, increased serum creatinine
Gastrointestinal Disorders:Abdominal pain, pancreatitis aggravated, salivary gland enlargement
Endocrine Reactions:Hyperthyroidism, hypothyroidism
Respiratory; Thoracic, and Mediastinal Disorders:Respiratory distress, respiratory failure, pulmonary edema, bronchospasm, laryngospasm, throat irritation, throat tightness, laryngeal edema, wheezing, chest discomfort, asthmatic attack
Skin and Subcutaneous Tissue Disorders:Contrast media reactions range from mild (e.g., pleomorphic rashes, drug eruption, erythema and skin discoloration, bulers, hyperhidrosis, angioedema, localized areas of edema) to severe: [e.g., Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN), bullous or exfoliative dermatitis, acute generalized exanthematous pustulosis (AGEP) and drug reaction with eosinophilia and systemic symptoms (DRESS)]
Oral Administration
Gastrointestinal Disorders:Dysphagia, abdominal pain
Body Cavity Administration
Gastrointestinal Disorders:Pancreatitis
Musculoskeletal and Connective Tissue Disorders:Arthritis (arthrography)
Hysterosalpingography: Injection of OMNIPAQUE for hysterosalpingography is associated with immediate, transient pain. Monitor injection pressure and volume instilled to minimize pain and to avoid disruptive distention of the uterus and fallopian tubes. Fluoroscopic monitoring is recommended.
Nervous system: Pain (49%), somnolence and fever each with an individual incidence of 3%.
Gastrointestinal system: Nausea (3%)
7 Drug Interactions
7.1 Drug-Drug Interactions
Metformin
In patients with renal impairment, metformin can cause lactic acidosis. Iodinated contrast agents appear to increase the risk of metformin-induced lactic acidosis, possibly as a result of worsening renal function. Stop metformin at the time of, or prior to, OMNIPAQUE administration in patients with an eGFR between 30 and 60 mL/min/1.73 m 2; in patients with a history of hepatic impairment, alcoholism or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure, and reinstitute metformin only after renal function is stable.
Radioactive Iodine
Administration of iodinated contrast agents may interfere with thyroid uptake of radioactive iodine (I-131 and I-123) and decrease therapeutic and diagnostic efficacy in patients with carcinoma of the thyroid. The decrease in efficacy lasts for 6 to 8 weeks.
Beta-adrenergic Blocking Agents
The use of beta-adrenergic blocking agents lowers the threshold for and increases the severity of contrast reactions and reduces the responsiveness of treatment of hypersensitivity reactions with epinephrine. Because of the risk of hypersensitivity reactions, use caution when administering OMNIPAQUE to patients taking beta-blockers.
Drugs that Lower Seizure Threshold
Drugs that lower seizure threshold, especially phenothiazine derivatives including those used for their antihistaminic or antinauseant properties, are not recommended for use with intrathecal administration of OMNIPAQUE.
CNS Active Drugs
Drugs such as monoamine oxidase (MAO) inhibitors, tricyclic antidepressants, CNS stimulants, psychoactive drugs described as analeptics, major tranquilizers, or antipsychotic drugs. Such medications should be discontinued at least 48 hours before myelography, should not be used for the control of nausea or vomiting during or after myelography, and should not be resumed for at least 24 hours post procedure. In non-elective procedures in patients on these drugs, consider prophylactic use of anticonvulsants.
7.2 Drug-Laboratory Test Interactions
Effect on Thyroid Tests
If iodine-containing isotopes are to be administered for the diagnosis of thyroid disease, the iodine-binding capacity of thyroid tissue may be reduced for up to 2 weeks after contrast medium administration. Thyroid function tests that do not depend on iodine estimation, e.g., T 3resin uptake or direct thyroxine assays, are not affected.
8 Use In Specific Populations
- Lactation: A lactating woman may pump and discard breast milk for 10 hours after OMNIPAQUE administration. (
8.2 )8.1 Pregnancy
Risk Summary
Hysterosalpingography is contraindicated in pregnant women due to the potential risk to the fetus from an intrauterine procedure [see Contraindications (4)]. There are no data with iohexol use in pregnant women to inform any drug-associated risks. Iohexol crosses the placenta and reaches fetal tissues in small amounts (see Data) . In animal reproduction studies, no developmental toxicity occurred with intravenous iohexol administration to rats and rabbits at doses up to 0.4 (rat) and 0.5 (rabbit) times the maximum recommended human intravenous dose ( see Data ).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Human Data
Literature reports show that intravenously administered iohexol crosses the placenta and is visualized in the digestive tract of exposed infants after birth.
Animal Data
Iohexol was neither embryotoxic nor teratogenic in either rats or rabbits at the following dose levels tested: 1.0, 2.0, 4.0 g iodine/kg in rats, administered intravenously to 3 groups of 25 dams once daily during days 6 through 15 of pregnancy; 0.3, 1.0, 2.5 g iodine/kg in rabbits, administered intravenously to 3 groups of 18 rabbits dosed once a day during days 6 through 18 of pregnancy.
8.2 Lactation
Risk Summary
Published literature reports that breast feeding after intravenous iohexol administration to the mother would result in the infant receiving an oral dose of approximately 0.7% of the maternal intravenous dose; however, lactation studies have not been conducted with oral, intrathecal, or intracavity administration of iohexol. There is no information on the effects of the drug on the breastfed infant or on milk production. Iodinated contrast agents are excreted unchanged in human milk in very low amounts with poor absorption from the gastrointestinal tract of a breastfed infant. Exposure to iohexol to a breastfed infant can be minimized by temporary discontinuation of breastfeeding (see Clinical Considerations). The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for OMNIPAQUE and any potential adverse effects on the breastfed infant from OMNIPAQUE or from the underlying maternal condition.
Clinical Considerations
Interruption of breastfeeding after exposure to iodinated contrast agents is not necessary because the potential exposure of the breastfed infant to iodine is small. However, a lactating woman may consider interrupting breastfeeding and pumping and discarding breast milk for 10 hours (approximately 5 elimination half-lives) after OMNIPAQUE administration to minimize drug exposure to a breastfed infant.
8.4 Pediatric Use
Intrathecal Use
The safety and effectiveness of OMNIPAQUE 180 have been established in pediatric patients 2 weeks to 17 years of age for myelography (lumbar, thoracic, cervical, total columnar) and for CT (myelography, cisternography). Use of OMNIPAQUE 180 is supported by controlled clinical studies in adults for myelography, in addition to clinical studies in pediatric patients undergoing myelography. The safety and effectiveness of OMNIPAQUE 180 have not been established for intrathecal use in patient pediatric patients less than 2 weeks of age. The safety and effectiveness of OMNIPAQUE 240 and 300 have not been established in pediatric patients for myelography (lumbar, thoracic, cervical, total columnar) and for CT (myelography, cisternography, or ventriculography).
Intravascular Use
Angiocardiography( Ventriculography, Pulmonary Arteriography, Venography, and Studies of the Collateral Arteries) and Aortography
The safety and effectiveness of OMNIPAQUE 300 have been established in pediatric patients from birth to 17 years of age for angiocardiography (ventriculography) and of OMNIPAQUE 350 in pediatric patients from birth to 17 years of age for angiocardiography (ventriculography, pulmonary arteriography, venography, and studies of the collateral arteries) and aortography. Use of OMNIPAQUE 300 and 350 is supported by controlled clinical studies in adults for angiocardiography and aortography, in addition to controlled clinical studies in pediatric patients undergoing angiocardiography, including aortography. The safety and effectiveness of OMNIPAQUE 300 have not been established in pediatric patients for aortography.
Intra-arterial Digital Subtraction Angiography, Intravenous Digital Subtraction Angiography, Cerebral Arteriography, or Peripheral Arteriography and Venography
The safety and effectiveness of OMNIPAQUE have not been established in pediatric patients for intra-arterial digital subtraction angiography, intravenous digital subtraction angiography, cerebral arteriography, or peripheral arteriography and venography.
CT of the Head and Body
The safety and effectiveness of OMNIPAQUE 240 and 300 have been established in pediatric patients from birth to 17 years of age for CT imaging of the head and body. Use of OMNIPAQUE 240 and 300 is supported by controlled clinical studies in adults for head and body CT, in addition to clinical studies in pediatric patients undergoing head CT and in 69 pediatric patients undergoing CT of the abdomen after oral administration of diluted OMNIPAQUE plus intravenous administration of OMNIPAQUE. The safety and effectiveness of OMNIPAQUE 350 have not been established in pediatric patients for CT imaging of the head and body.
Urography
The safety and effectiveness of OMNIPQUE 300 have been established in pediatric patients from birth to 17 years of age for urography. Use of OMNIPAQUE 300 is supported by controlled clinical studies in adults for urography, in addition to controlled clinical studies in pediatric patients undergoing urography and clinical safety data in pediatric patients down to birth.
Oral or Rectal Use
Undiluted OMNIPAQUE Injection
The safety and effectiveness of OMNIPAQUE 180, 240, and 300 administered orally and rectally have been established in pediatric patients, from birth to 17 years of age for examination of the GI tract. Use of OMNIPAQUE 180, 240, and 300 administered orally and rectally is supported by controlled studies in adults for examination of the GI tract, in addition to clinical studies in pediatric patients undergoing examination of the GI tract.
Oral Use in Conjunction with Intravenous Use
Diluted OMNIPAQUE Injection
The safety and effectiveness of OMNIPAQUE injection diluted to concentrations from 9 to 21 mg iodine/mL administered orally in conjunction with OMNIPAQUE injection administered intravenously for CT of the abdomen have been established in pediatric patients from birth to 17 years of age. Use is supported by clinical trials in adults, in addition to clinical studies in 69 pediatric patients undergoing CT of the abdomen after oral administration of diluted OMNIPAQUE plus intravenous administration of OMNIPAQUE.
OMNIPAQUE Oral Solution
The safety and effectiveness of OMNIPAQUE oral solution 9 and 12 administered orally in conjunction with OMNIPAQUE injection administered intravenously for CT of the abdomen in pediatric patients have been established in pediatric patients from birth to 17 years of age. Use is supported by the data establishing safety and effectiveness for OMNIPAQUE injection diluted and administered orally in conjunction with OMNIPAQUE injection administered intravenously for CT of the abdomen in pediatric patients.
Intraarticular Use
The safety and effectiveness of OMNIPAQUE have not been established in pediatric patients for arthrography.
Body Cavity Use
OMNIPAQUE 240, 300, 350 diluted to concentrations from 50 mg iodine/mL to 100 mg iodine/mL is indicated for use in pediatric patients from birth to 17 years of age for voiding cystourethrography (VCU). The use for voiding cystourethrography is supported by clinical studies in 51 pediatric patients undergoing VCU. The safety and effectiveness of OMNIPAQUE have not been established in pediatric patients for ERCP, herniography, or hysterosalpingography.
In general, the frequency of adverse reactions in pediatric patients was similar to that seen in adults [see Adverse Reactions (6.1)] . Pediatric patients at higher risk of experiencing adverse events during contrast-medium administration may include those having asthma, a sensitivity to medication and/or allergens, congestive heart failure, a serum creatinine greater than 1.5 mg/dL or those less than 12 months of age.
Thyroid function tests indicative of thyroid dysfunction, characterized by hypothyroidism or transient thyroid suppression have been reported following iodinated contrast media administration in pediatric patients, including term and preterm neonates. Some patients were treated for hypothyroidism. After exposure to iodinated contrast media, individualize thyroid function monitoring in pediatric patients 0 to 3 years of age based on underlying risk factors, especially in term and preterm neonates [see Warnings and Precautions (5.9)and Adverse Reactions (6.2)] .
8.5 Geriatric Use
In clinical studies of OMNIPAQUE for CT, 52/299 (17%) of patients were 70 and over. No overall differences in safety were observed between these patients and younger patients. Other reported clinical experience has not identified differences in response between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. In general, dose selection for an elderly patient should be cautious usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
10 Overdosage
10.1 Intravascular Administration
The adverse effects of overdosage are life-threatening and affect mainly the pulmonary and cardiovascular systems. The symptoms included: cyanosis, bradycardia, acidosis, pulmonary hemorrhage, convulsions, coma, and cardiac arrest. Treatment of an overdosage is directed toward the support of all vital functions, and prompt institution of symptomatic therapy. Iohexol displays a low affinity for serum or plasma proteins and is poorly bound to serum albumin and can be dialyzed.
11 Description
11.1 Chemical Characteristics
OMNIPAQUE (iohexol) injection is a nonionic, x-ray or radiographic contrast medium for intrathecal, intravenous, oral, rectal and body cavity use. OMNIPAQUE oral solution is for oral use only.
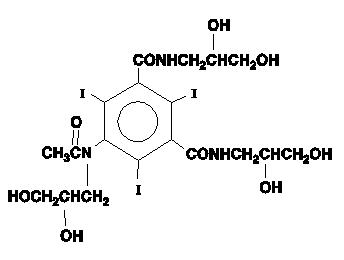
OMNIPAQUE injection and OMNIPAQUE oral solution are both provided as sterile, pyrogen-free and gluten-free solutions. OMNIPAQUE injection and OMNIPAQUE oral solution are colorless to pale yellow solutions. The chemical name of iohexol is Bis(2,3-dihydroxypropyl)-5-[ N-(2,3-dihydroxypropyl)-acetamido]-2,4,6- triiodoisophthalamide with a molecular weight of 821.14 (iodine content 46.36%). Iohexol has the following structural formula:
OMNIPAQUE injection is available in five strengths:
- OMNIPAQUE 140 mg iodine/mL (302 mg of iohexol/mL): Each mL contains 140 mg organically bound iodine, 1.21 mg tromethamine and 0.1 mg edetate calcium disodium
- OMNIPAQUE 180 mg iodine/mL (388 mg of iohexol/mL): Each mL contains 180 mg organically bound iodine, 1.21 mg tromethamine and 0.1 mg edetate calcium disodium
- OMNIPAQUE 240 mg iodine/mL (518 mg of iohexol/mL): Each mL contains 240 mg organically bound iodine, 1.21 mg tromethamine and 0.1 mg edetate calcium disodium
- OMNIPAQUE 300 mg iodine/mL (647 mg of iohexol/mL): Each mL contains 300 mg organically bound iodine, 1.21 mg tromethamine and 0.1 mg edetate calcium disodium
- OMNIPAQUE 350 mg iodine/mL (755 mg of iohexol/mL): Each mL contains 350 mg organically bound iodine, 1.21 mg tromethamine and 0.1 mg edetate calcium disodium
OMNIPAQUE oral solution is available in two strengths:
- OMNIPAQUE oral solution 9 mg iodine/mL (19 mg of iohexol/mL): Each mL contains 9 mg organically bound iodine, 1.21 mg tromethamine and 0.1 mg edetate calcium disodium
- OMNIPAQUE oral solution 12 mg iodine/mL (26 mg of iohexol/mL): Each mL contains 12 mg organically bound iodine, 1.21 mg tromethamine and 0.1 mg edetate calcium disodium
The pH is adjusted between 6.8 and 7.7 with hydrochloric acid or sodium hydroxide. OMNIPAQUE injection and OMNIPAQUE oral solution are sterilized by autoclaving and contain no preservatives.
11.2 Physical Characteristics
OMNIPAQUE injection and OMNIPAQUE oral solution have the following physical properties:
Presentation Concentration (mg iodine/mL) Osmolality By vapor-pressure osmometry. (mOsmol/kg water)Absolute Viscosity (cP) Specific Gravity 20°C 37°C 37°C OMNIPAQUE 140 140 322 2.3 1.5 1.164 OMNIPAQUE 180 180 408 3.1 2.0 1.209 OMNIPAQUE 240 240 520 5.8 3.4 1.280 OMNIPAQUE 300 300 672 11.8 6.3 1.349 OMNIPAQUE 350 350 844 20.4 10.4 1.406 OMNIPAQUE oral solution 9 9 38 1.1 0.8 1.011 OMNIPAQUE oral solution 12 12 45 1.1 0.8 1.014
OMNIPAQUE 140, OMNIPAQUE 180, OMNIPAQUE 240, OMNIPAQUE 300, and OMNIPAQUE 350 have osmolalities from approximately 1.1 to 3.0 times that of plasma (285 mOsmol/kg water) or cerebrospinal fluid (301 mOsmol/kg water) as shown in the above table and are hypertonic under conditions of use.
OMNIPAQUE oral solution 9 and OMNIPAQUE oral solution 12 are hypotonic under conditions of use (see tableabove).
12 Clinical Pharmacology
12.1 Mechanism of Action
The iodine atoms in iohexol provide attenuation of X-rays in direct proportion to the concentration of iohexol. Since concentration changes over time, iohexol provides time-dependent image contrast which may assist in visualizing body structures.
12.2 Pharmacodynamics
Intrathecal Administration
The initial concentration and volume of the contrast medium, in conjunction with patient manipulation and the volume of cerebrospinal fluid (CSF) into which the contrast medium is placed, will determine the extent of the contrast that can be achieved. Following intrathecal injection in conventional radiography, OMNIPAQUE 180, 240, and 300 will continue to provide contrast for at least 30 minutes. Slow diffusion of iohexol takes place throughout the CSF with subsequent absorption into the bloodstream. At approximately 1 hour following injection, contrast will no longer be sufficient for conventional myelography.
After administration into the lumbar subarachnoid space, computerized tomography shows the presence of contrast medium in the thoracic region in about 1 hour, in the cervical region in about 2 hours, and in the basal cisterns in 3 to 4 hours.
Intravascular Administration
Following intravascular administration of OMNIPAQUE, the degree of contrast enhancement is directly related to the iodine concentration of an administered dose; peak iodine blood concentrations occur immediately (15 seconds to 120 seconds) following rapid intravenous injection. The time to maximum contrast enhancement can vary, depending on the organ, from the time that peak blood iodine concentrations are reached to one hour after intravenous bolus administration. When a delay between peak blood iodine concentrations and peak contrast is present, it suggests that radiographic contrast enhancement is at least in part dependent on the accumulation of iodine containing agent within the lesion and outside the blood pool.
Oral Administration
Orally administered OMNIPAQUE produces visualization of the gastrointestinal tract. Less than 1% of orally administered iohexol is recovered in the urine, suggesting minimal amounts are absorbed from the normal gastrointestinal tract. This amount may increase in the presence of bowel perforation or bowel obstruction.
Intraarticular Administration
Visualization of the joint spaces can be accomplished by direct injection of contrast medium. For intraarticular cavities, the injected iohexol is absorbed into the surrounding tissue and subsequently absorbed into systemic circulation.
Body Cavity Administration
For most body cavities, the injected iohexol is absorbed into the surrounding tissue and subsequently absorbed into systemic circulation. Examinations of the uterus (hysterosalpingography) and bladder (voiding cystourethrography) involve the almost immediate drainage of contrast medium from the cavity upon conclusion of the radiographic procedure.
12.3 Pharmacokinetics
Following the intravenous administration of iohexol (between 500 mg iodine/kg to 1500 mg iodine/kg) to 16 adult human subjects, apparent first-order terminal elimination half-life was 12.6 hrs and total body clearance was 131 (98 to 165) mL/min. Clearance was not dose dependent.
Absorption
As evidenced by the amount recovered in urine, <1% of orally administered iohexol is absorbed from the normal gastrointestinal tract. This amount may increase in the presence of bowel perforation or bowel obstruction.
Distribution
In 16 adult subjects (receiving between 500 mg iodine/kg to 1500 mg iodine/kg intravenous iohexol) the plasma volume of distribution was165 (108 to 219) mL/kg.
In five adult patients receiving 16 mL to 18 mL of OMNIPAQUE (180 mg iodine/mL) by lumbar intrathecal injection the plasma volume of distribution was 559 (350 to 849) mL/kg.
Elimination
Metabolism
No significant metabolism, deiodination or biotransformation occurs.
Excretion
Following intravascular or intrathecal administration, iohexol is excreted unchanged by glomerular filtration. Approximately 90% of the intravenously injected iohexol dose is excreted within the first 24 hours. Following intravascular administration, peak urine concentration occurs in the first hour after injection.
13 Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been performed with iohexol to evaluate carcinogenic potential. Iohexol was not genotoxic in a series of studies, including the Ames test, the mouse lymphoma TK locus forward mutation assay, and a mouse micronucleus assay. Iohexol did not impair the fertility of male or female rats when repeatedly administered at intravenous dosages up to 4 g iodine/kg.
16 How Supplied/storage And Handling
16.1 How Supplied
Volume/Concentration Configuration NDC OMNIPAQUE 140 (140 mg iodine/mL) – Boxes of 10 50 mL + PLUSPAK™ (polymer bottle) 0407-1401-52 OMNIPAQUE 180 (180 mg iodine/mL) – Boxes of 10 10 mL Glass Vial 0407-1411-10 OMNIPAQUE 240 (240 mg iodine/mL) – Boxes of 10 10 mL Glass Vial 0407-1412-10 20 mL Glass Vial 0407-1412-20 50 mL + PLUSPAK™ (polymer bottle) 0407-1412-30 100 mL + PLUSPAK™ (polymer bottle) 0407-1412-33 OMNIPAQUE 300 (300 mg iodine/mL) – Boxes of 10 10 mL Glass Vial 0407-1413-10 30 mL fill in 50 mL + PLUSPAK™ (polymer bottle) 0407-1413-59 50 mL + PLUSPAK™ (polymer bottle) 0407-1413-61 100 mL + PLUSPAK™ (polymer bottle) 0407-1413-63 125 mL fill in 150 mL Glass Bottle 0407-1413-53 150 mL fill in 200 mL + PLUSPAK™ (polymer bottle) 0407-1413-65 OMNIPAQUE 350 (350 mg iodine/mL) – Boxes of 10 50 mL + PLUSPAK™ (polymer bottle) 0407-1414-89 75 mL fill in 100 mL + PLUSPAK™ (polymer bottle) 0407-1414-90 100 mL + PLUSPAK™ (polymer bottle) 0407-1414-91 125 mL fill in 150 mL Glass Bottle 0407-1414-76 150 mL fill in 200 mL + PLUSPAK™ (polymer bottle) 0407-1414-93 200 mL + PLUSPAK™ (polymer bottle) 0407-1414-94 OMNIPAQUE Oral Solution 9 (9 mg iodine/mL) – Boxes of 10 500 mL + PLUSPAK™ (polymer bottle) 0407-1415-09 OMNIPAQUE Oral Solution 12 (12 mg iodine/mL) – Boxes of 10 500 mL + PLUSPAK™ (polymer bottle) 0407-1416-12
The container closure system components (bottle, vial, stopper and cap) of OMNIPAQUE injection and OMNIPAQUE oral solution are not made with natural rubber latex.
16.2 Storage and Handling
Protect OMNIPAQUE glass vials and bottles and + PLUSPAK™ polymer bottles from light. Do not freeze. Discard any product that is inadvertently frozen, as freezing may compromise the closure integrity of the immediate container.
OMNIPAQUE Injection 140, 180, 240, 300 and 350
Store at controlled room temperature, 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. May be stored in a contrast media warmer for up to one month at 36° to 38°C (96.8° to 100.4°F).
OMNIPAQUE Oral Solution 9 and 12
Store between 0° and 30°C (32° to 86°F).
17 Patient Counseling Information
Hypersensitivity Reactions
Advise the patient concerning the risk of hypersensitivity reactions that can occur both during and after OMNIPAQUE administration. Advise the patient to report any signs or symptoms of hypersensitivity reactions during the procedure and to seek immediate medical attention for any signs or symptoms experienced after discharge [see Warnings and Precautions (5.3)]
Advise patients to inform their physician if they develop a rash after receiving OMNIPAQUE [see Warnings and Precautions (5.12)].
Contrast-Induced Acute Kidney Injury
Advise the patient concerning appropriate hydration to decrease the risk of contrast-induced acute kidney injury [see Warnings and Precautions (5.4)] .
Extravasation
If extravasation occurs during injection, advise patients to seek medical care for progression of symptoms [see Warnings and Precautions (5.7)].
LACTATION SECTION
Lactation
Advise a lactating woman that interruption of breastfeeding is not necessary. However, to avoid any exposure, a lactating woman may consider pumping and discarding breast milk for 10 hours after OMNIPAQUE administration [ see Use in Specific Populations (8.2)] .
Thyroid Dysfunction
Advise parents/caregivers about the risk of developing thyroid dysfunction after OMNIPAQUE administration. Advise parents/caregivers about when to seek medical care for their child to monitor for thyroid function [see Warnings and Precautions (5.9)].
Distributed by GE Healthcare Inc., Marlborough, MA 01752 U.S.A.
Product of Norwegian Origin.
OMNIPAQUE is a trademark of GE HealthCare or one of its subsidiaries. GE is a trademark of General Electric Company used under trademark license.
© 2023 GE HealthCare
Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension, USP, 6 mg/mL
30 mg/5 mL (6 mg/mL)
Description
Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension is a sterile aqueous suspension containing 3 mg per milliliter betamethasone, as betamethasone sodium phosphate, and 3 mg per milliliter betamethasone acetate. Inactive ingredients per mL: 7.1 mg dibasic sodium phosphate anhydrous; 3.4 mg monobasic sodium phosphate monohydrate; 0.1 mg edetate disodium; and 0.2 mg benzalkonium chloride as a preservative. The pH is adjusted to between 6.8 and 7.2.
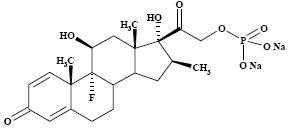
The formula for betamethasone sodium phosphate is C 22H 28FNa 2O 8P and it has a molecular weight of 516.40. Chemically, it is 9-Fluoro-11β,17,21-trihydroxy-16β-methylpregna-1,4-diene-3,20-dione 21-(disodium phosphate).
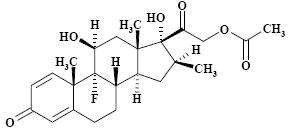
The formula for betamethasone acetate is C 24H 31FO 6and it has a molecular weight of 434.50. Chemically, it is 9-Fluoro-11β,17,21-trihydroxy-16β-methylpregna-1,4-diene-3,20-dione 21-acetate.
The chemical structures for betamethasone sodium phosphate and betamethasone acetate are as follows:
Betamethasone sodium phosphate is a white to practically white, odorless powder, and is hygroscopic. It is freely soluble in water and in methanol, but is practically insoluble in acetone and in chloroform.
Betamethasone acetate is a white to creamy white, odorless powder that sinters and resolidifies at about 165ÂşC, and remelts at about 200ÂşC to 220ÂşC with decomposition. It is practically insoluble in water, but freely soluble in acetone, and is soluble in alcohol and in chloroform.
Clinical Pharmacology
Glucocorticoids, naturally occurring and synthetic, are adrenocortical steroids that are readily absorbed from the gastrointestinal tract.
Naturally occurring glucocorticoids (hydrocortisone and cortisone), which also have salt-retaining properties, are used as replacement therapy in adrenocortical deficiency states. Their synthetic analogs are primarily used for their anti-inflammatory effects in disorders of many organ systems. A derivative of prednisolone, betamethasone has a 16β-methyl group that enhances the anti-inflammatory action of the molecule and reduces the sodium- and water-retaining properties of the fluorine atom bound at carbon 9.
Betamethasone sodium phosphate, a soluble ester, provides prompt activity, while betamethasone acetate is only slightly soluble and affords sustained activity.
Indications And Usage
When oral therapy is not feasible, the intramuscular useof Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension is indicated as follows:
Allergic StatesControl of severe or incapacitating allergic conditions intractable to adequate trials of conventional treatment in asthma, atopic dermatitis, contact dermatitis, drug hypersensitivity reactions, perennial or seasonal allergic rhinitis, serum sickness, transfusion reactions.
Dermatologic DiseasesBullous dermatitis herpetiformis, exfoliative erythroderma, mycosis fungoides, pemphigus, severe erythema multiforme (Stevens-Johnson syndrome).
Endocrine DisordersCongenital adrenal hyperplasia, hypercalcemia associated with cancer, nonsuppurative thyroiditis.
Hydrocortisone or cortisone is the drug of choice in primary or secondary adrenocortical insufficiency. Synthetic analogs may be used in conjunction with mineralocorticoids where applicable; in infancy mineralocorticoid supplementation is of particular importance.
Gastrointestinal DiseasesTo tide the patient over a critical period of the disease in regional enteritis and ulcerative colitis.
Hematologic DisordersAcquired (autoimmune) hemolytic anemia, Diamond-Blackfan anemia, pure red cell aplasia, selected cases of secondary thrombocytopenia.
MiscellaneousTrichinosis with neurologic or myocardial involvement, tuberculous meningitis with subarachnoid block or impending block when used with appropriate antituberculous chemotherapy.
Neoplastic DiseasesFor palliative management of leukemias and lymphomas.
Nervous SystemAcute exacerbations of multiple sclerosis; cerebral edema associated with primary or metastatic brain tumor or craniotomy.
Ophthalmic DiseasesSympathetic ophthalmia, temporal arteritis, uveitis and ocular inflammatory conditions unresponsive to topical corticosteroids.
Renal DiseasesTo induce diuresis or remission of proteinuria in idiopathic nephrotic syndrome or that due to lupus erythematosus.
Respiratory DiseasesBerylliosis, fulminating or disseminated pulmonary tuberculosis when used concurrently with appropriate antituberculous chemotherapy, idiopathic eosinophilic pneumonias, symptomatic sarcoidosis.
Rheumatic DisordersAs adjunctive therapy for short-term administration (to tide the patient over an acute episode or exacerbation) in acute gouty arthritis; acute rheumatic carditis; ankylosing spondylitis; psoriatic arthritis; rheumatoid arthritis, including juvenile rheumatoid arthritis (selected cases may require low-dose maintenance therapy). For the treatment of dermatomyositis, polymyositis, and systemic lupus erythematosus.
The intra-articular or soft tissue administrationof Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension is indicated as adjunctive therapy for short-term administration (to tide the patient over an acute episode or exacerbation) in acute gouty arthritis, acute and subacute bursitis, acute nonspecific tenosynovitis, epicondylitis, rheumatoid arthritis, synovitis of osteoarthritis.
The intralesional administrationof Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension is indicated for alopecia areata; discoid lupus erythematosus; keloids; localized hypertrophic, infiltrated, inflammatory lesions of granuloma annulare, lichen planus, lichen simplex chronicus (neurodermatitis), and psoriatic plaques; necrobiosis lipoidica diabeticorum.
Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension may also be useful in cystic tumors of an aponeurosis or tendon (ganglia).
Contraindications
Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension is contraindicated in patients who are hypersensitive to any components of this product (see DESCRIPTION ). Intramuscular corticosteroid preparations are contraindicated for idiopathic thrombocytopenic purpura.
Warnings
Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension should not be administered intravenously.
Serious Neurologic Adverse Reactions with Epidural Administration
Serious neurologic events, some resulting in death, have been reported with epidural injection of corticosteroids. Specific events reported include, but are not limited to, spinal cord infarction, paraplegia, quadriplegia, cortical blindness, and stroke. These serious neurologic events have been reported with and without use of fluoroscopy. The safety and effectiveness of epidural administration of corticosteroids have not been established, and corticosteroids are not approved for this use.
General
Rare instances of anaphylactoid/anaphylactic reactions with a possibility of shock have occurred in patients receiving parenteral corticosteroid therapy (see ADVERSE REACTIONS). Use caution in patients who have a history of allergic reaction to corticosteroids.
In patients on corticosteroid therapy subjected to any unusual stress, hydrocortisone or cortisone is the drug of choice as a supplement during and after the event.
Cardio-Renal
Average and large doses of corticosteroids can cause elevation of blood pressure, salt and water retention, and increased excretion of potassium. These effects are less likely to occur with the synthetic derivatives except when used in large doses. Dietary salt restriction and potassium supplementation may be necessary. All corticosteroids increase calcium excretion.
Literature reports suggest an apparent association between use of corticosteroids and left ventricular free wall rupture after a recent myocardial infarction; therefore, therapy with corticosteroids should be used with great caution in these patients.
Endocrine
Corticosteroids can produce reversible hypothalamic pituitary adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency after withdrawal of treatment.
Metabolic clearance of corticosteroids is decreased in hypothyroid patients and increased in hyperthyroid patients. Changes in thyroid status of the patient may necessitate adjustment in dosage.
Infections
Patients who are on corticosteroids are more susceptible to infections than are healthy individuals. There may be decreased resistance and inability to localize infection when corticosteroids are used. Infection with any pathogen (viral, bacterial, fungal, protozoan, or helminthic) in any location of the body may be associated with the use of corticosteroids alone or in combination with other immunosuppressive agents. These infections may be mild to severe. With increasing doses of corticosteroids, the rate of occurrence of infectious complications increases. Corticosteroids may also mask some signs of current infection.
Corticosteroids may exacerbate systemic fungal infections and therefore should not be used in the presence of such infections unless they are needed to control drug reactions. There have been cases reported in which concomitant use of amphotericin B and hydrocortisone was followed by cardiac enlargement and congestive heart failure (see PRECAUTIONS, Drug Interactions, Amphotericin B Injection and Potassium-Depleting Agentssection).
Latent disease may be activated or there may be an exacerbation of intercurrent infections due to pathogens, including those caused by Amoeba, Candida, Cryptococcus, Mycobacterium, Nocardia, Pneumocystis, and Toxoplasma. It is recommended that latent amebiasis or active amebiasis be ruled out before initiating corticosteroid therapy in any patient who has spent time in the tropics or in any patient with unexplained diarrhea.
Similarly, corticosteroids should be used with great care in patients with known or suspected Strongyloides (threadworm) infestation. In such patients, corticosteroid-induced immunosuppression may lead to Strongyloides hyperinfection and dissemination with widespread larval migration, often accompanied by severe enterocolitis and potentially fatal gram-negative septicemia.
Corticosteroids should not be used in cerebral malaria.
The use of corticosteroids in active tuberculosis should be restricted to those cases of fulminating or disseminated tuberculosis in which the corticosteroid is used for the management of the disease in conjunction with an appropriate antituberculous regimen.
If corticosteroids are indicated in patients with latent tuberculosis or tuberculin reactivity, close observation is necessary as reactivation of the disease may occur. During prolonged corticosteroid therapy, these patients should receive chemoprophylaxis.
Administration of live or live, attenuated vaccines is contraindicated in patients receiving immunosuppressive doses of corticosteroids. Killed or inactivated vaccines may be administered. However, the response to such vaccines cannot be predicted.Immunization procedures may be undertaken in patients who are receiving corticosteroids as replacement therapy, e.g., for Addison’s disease.
Chickenpox and measles can have a more serious or even fatal course in pediatric and adult patients on corticosteroids. In pediatric and adult patients who have not had these diseases, particular care should be taken to avoid exposure. The contribution of the underlying disease and/or prior corticosteroid treatment to the risk is also not known. If exposed to chickenpox, prophylaxis with varicella zoster immune globulin (VZIG) may be indicated. If exposed to measles, prophylaxis with immunoglobulin (IG) may be indicated. (See the respective package inserts for complete VZIG and IG prescribing information.) If chickenpox develops, treatment with antiviral agents should be considered.
Neurologic
Reports of severe medical events have been associated with the intrathecal route of administration (see ADVERSE REACTIONS, Gastrointestinal and Neurologic/Psychiatric sections).
Results from one multicenter, randomized, placebo-controlled study with methylprednisolone hemisuccinate, an intravenous corticosteroid, showed an increase in early mortality (at 2 weeks) and late mortality (at 6 months) in patients with cranial trauma who were determined not to have other clear indications for corticosteroid treatment. High doses of corticosteroids, including Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension, should not be used for the treatment of traumatic brain injury.
Ophthalmic
Use of corticosteroids may produce posterior subcapsular cataracts, increased intraocular pressure, glaucoma with possible damage to the optic nerves, and may enhance the establishment of secondary ocular infections due to bacteria, fungi, or viruses. Consider referral to an ophthalmologist for patients who develop ocular symptoms or use corticosteroid-containing products for more than 6 weeks. The use of oral corticosteroids is not recommended in the treatment of optic neuritis and may lead to an increase in the risk of new episodes. Corticosteroids should not be used in active ocular herpes simplex.
Precautions
General
This product, like many other steroid formulations, is sensitive to heat. Therefore, it should not be autoclaved when it is desirable to sterilize the exterior of the vial.
The lowest possible dose of corticosteroid should be used to control the condition under treatment. When reduction in dosage is possible, the reduction should be gradual.
Since complications of treatment with glucocorticoids are dependent on the size of the dose and the duration of treatment, a risk/benefit decision must be made in each individual case as to dose and duration of treatment and as to whether daily or intermittent therapy should be used.
Kaposi’s sarcoma has been reported to occur in patients receiving corticosteroid therapy, most often for chronic conditions. Discontinuation of corticosteroids may result in clinical improvement.
Cardio-Renal
As sodium retention with resultant edema and potassium loss may occur in patients receiving corticosteroids, these agents should be used with caution in patients with congestive heart failure, hypertension, or renal insufficiency.
Endocrine
Drug-induced secondary adrenocortical insufficiency may be minimized by gradual reduction of dosage. This type of relative insufficiency may persist for months after discontinuation of therapy. Therefore, in any situation of stress occurring during that period, naturally occurring glucocorticoids (hydrocortisone cortisone), which also have salt-retaining properties, rather than betamethasone, are the appropriate choices as replacement therapy in adrenocortical deficiency states.
Gastrointestinal
Steroids should be used with caution in active or latent peptic ulcers, diverticulitis, fresh intestinal anastomoses, and nonspecific ulcerative colitis, since they may increase the risk of a perforation.
Signs of peritoneal irritation following gastrointestinal perforation in patients receiving corticosteroids may be minimal or absent.
There is an enhanced effect of corticosteroids in patients with cirrhosis.
Intra-Articular and Soft Tissue Administration
Intra-articular injected corticosteroids may be systemically absorbed.
Appropriate examination of any joint fluid present is necessary to exclude a septic process.
A marked increase in pain accompanied by local swelling, further restriction of joint motion, fever, and malaise are suggestive of septic arthritis. If this complication occurs and the diagnosis of sepsis is confirmed, appropriate antimicrobial therapy should be instituted.
Injection of a steroid into an infected site is to be avoided. Local injection of a steroid into a previously injected joint is not usually recommended.
Corticosteroid injection into unstable joints is generally not recommended.
Intra-articular injection may result in damage to joint tissues (see ADVERSE REACTIONS, Musculoskeletal section).
Musculoskeletal
Corticosteroids decrease bone formation and increase bone resorption both through their effect on calcium regulation (i.e., decreasing absorption and increasing excretion) and inhibition of osteoblast function. This, together with a decrease in the protein matrix of the bone secondary to an increase in protein catabolism, and reduced sex hormone production, may lead to inhibition of bone growth in pediatric patients and the development of osteoporosis at any age. Special consideration should be given to patients at increased risk of osteoporosis (i.e., postmenopausal women) before initiating corticosteroid therapy.
Neuro-Psychiatric
Although controlled clinical trials have shown corticosteroids to be effective in speeding the resolution of acute exacerbations of multiple sclerosis, they do not show that they affect the ultimate outcome or natural history of the disease. The studies do show that relatively high doses of corticosteroids are necessary to demonstrate a significant effect (see DOSAGE AND ADMINISTRATION).
An acute myopathy has been observed with the use of high doses of corticosteroids, most often occurring in patients with disorders with neuromuscular transmission (e.g., myasthenia gravis), or in patients receiving concomitant therapy of neuromuscular blocking drugs (eg, pancuronium). This acute myopathy is generalized, may involve ocular and respiratory muscles, and may result in quadriparesis. Elevation of creatinine kinase may occur. Clinical improvement or recovery after stopping corticosteroids may require weeks to years.
Psychic derangements may appear when corticosteroids are used, ranging from euphoria, insomnia, mood swings, personality changes, and severe depression to frank psychotic manifestations. Also, existing emotional instability or psychotic tendencies may be aggravated by corticosteroids.
Information for Patients
Patients should be warned not to discontinue the use of corticosteroids abruptly or without medical supervision, to advise any medical attendants that they are taking corticosteroids and to seek medical advice at once should they develop fever or other signs of infection.
Persons who are on corticosteroids should be warned to avoid exposure to chickenpox or measles. Patients should also be advised that if they are exposed, medical advice should be sought without delay.
Drug Interactions
Aminoglutethimide may lead to a loss of corticosteroid-induced adrenal suppression.
When corticosteroids are administered concomitantly with potassium-depleting agents (ie, amphotericin B, diuretics), patients should be observed closely for development of hypokalemia. There have been cases reported in which concomitant use of amphotericin B and hydrocortisone was followed by cardiac enlargement and congestive heart failure.
Macrolide antibiotics have been reported to cause a significant decrease in corticosteroid clearance.
Concomitant use of anticholinesterase agents and corticosteroids may produce severe weakness in patients with myasthenia gravis. If possible, anticholinesterase agents should be withdrawn at least 24 hours before initiating corticosteroid therapy.
Coadministration of corticosteroids and warfarin usually results in inhibition of response to warfarin, although there have been some conflicting reports. Therefore, coagulation indices should be monitored frequently to maintain the desired anticoagulant effect.
Because corticosteroids may increase blood glucose concentrations, dosage adjustments of antidiabetic agents may be required.
Serum concentrations of isoniazid may be decreased.
Cholestyramine may increase the clearance of corticosteroids.
Increased activity of both cyclosporine and corticosteroids may occur when the two are used concurrently. Convulsions have been reported with this concurrent use.
Patients on digitalis glycosides may be at increased risk of arrhythmias due to hypokalemia.
Estrogens may decrease the hepatic metabolism of certain corticosteroids, thereby increasing their effect.
Drugs which induce hepatic microsomal drug-metabolizing enzyme activity may enhance the metabolism of corticosteroids and require that the dosage of the corticosteroid be increased.
Corticosteroids (including betamethasone) are metabolized by CYP3A4.
Ketoconazole has been reported to decrease the metabolism of certain corticosteroids by up to 60%, leading to an increased risk of corticosteroid side effects.
Coadministration with other strong CYP3A4 inhibitors (e.g., itraconazole, clarithromycin, ritonavir, cobicistat-containing products) may lead to increased exposures of corticosteroids and therefore the potential for increased risk of systemic corticosteroid side effects.
Consider the benefit of coadministration versus the potential risk of systemic corticosteroid effects, in which case patients should be monitored for systemic corticosteroid side effects.
Concomitant use of aspirin (or other nonsteroidal anti-inflammatory agents) and corticosteroids increases the risk of gastrointestinal side effects. Aspirin should be used cautiously in conjunction with corticosteroids in hypoprothrombinemia. The clearance of salicylates may be increased with concurrent use of corticosteroids.
Corticosteroids may suppress reactions to skin tests.
Patients on prolonged corticosteroid therapy may exhibit a diminished response to toxoids and live or inactivated vaccines due to inhibition of antibody response. Corticosteroids may also potentiate the replication of some organisms contained in live attenuated vaccines. Route administration of vaccines or toxoids should be deferred until corticosteroid therapy is discontinued if possible (see WARNINGS, Infections, Vaccinationsection).
Carcinogenesis, Mutagenesis, Impairment of Fertility
No adequate studies have been conducted in animals to determine whether corticosteroids have a potential for carcinogenesis or mutagenesis.
Steroids may increase or decrease motility and number of spermatozoa in some patients.
Pregnancy
Corticosteroids have been shown to be teratogenic in many species when given in doses equivalent to the human dose. Animal studies in which corticosteroids have been given to pregnant mice, rats, and rabbits have yielded an increased incidence of cleft palate in the offspring. There are no adequate and well-controlled studies in pregnant women. Corticosteroids should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Infants born to mothers who have received corticosteroids during pregnancy should be carefully observed for signs of hypoadrenalism.
Nursing Mothers
Systemically administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. Caution should be exercised when corticosteroids are administered to a nursing woman.
Pediatric Use
The efficacy and safety of corticosteroids in the pediatric population are based on the well-established course of effect of corticosteroids, which is similar in pediatric and adult populations. Published studies provide evidence of efficacy and safety in pediatric patients for the treatment of nephrotic syndrome (> 2 years of age), and aggressive lymphomas and leukemias (> 1 month of age). Other indications for pediatric use of corticosteroids, e.g., severe asthma and wheezing, are based on adequate and well-controlled trials conducted in adults, on the premises that the course of the diseases and their pathophysiology are considered to be substantially similar in both populations.
The adverse effects of corticosteroids in pediatric patients are similar to those in adults (see ADVERSE REACTIONS). Like adults, pediatric patients should be carefully observed with frequent measurements of blood pressure, weight, height, intraocular pressure, and clinical evaluation for the presence of infection, psychosocial disturbances, thromboembolism, peptic ulcers, cataracts, and osteoporosis. Pediatric patients who are treated with corticosteroids by any route, including systemically administered corticosteroids, may experience a decrease in their growth velocity. This negative impact of corticosteroids on growth has been observed at low systemic doses and in the absence of laboratory evidence of HPA axis suppression (i.e., cosyntropin stimulation and basal cortisol plasma levels). Growth velocity may therefore be a more sensitive indicator of systemic corticosteroid exposure in pediatric patients than some commonly used tests of HPA axis function. The linear growth of pediatric patients treated with corticosteroids should be monitored, and the potential growth effects of prolonged treatment should be weighed against clinical benefits obtained and the availability of treatment alternatives. In order to minimize the potential growth effects of corticosteroids, pediatric patients should be titrated to the lowest effective dose.
Geriatric Use
No overall differences in safety or effectiveness were observed between elderly subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and young patients, but greater sensitivity of some older individuals cannot be ruled out.
Adverse Reactions
(uled alphabetically, under each subsection)
Allergic Reactions
Anaphylactoid reaction, anaphylaxis, angioedema.
Cardiovascular
Bradycardia, cardiac arrest, cardiac arrhythmias, cardiac enlargement, circulatory collapse, congestive heart failure, fat embolism, hypertension, hypertrophic cardiomyopathy in premature infants, myocardial rupture following recent myocardial infarction (see WARNINGS), pulmonary edema, syncope, tachycardia, thromboembolism, thrombophlebitis, vasculitis.
Dermatologic
Acne, allergic dermatitis, cutaneous and subcutaneous atrophy, dry scaly skin, ecchymoses and petechiae, edema, erythema, hyperpigmentation, hypopigmentation, impaired wound healing, increased sweating, rash, sterile abscess, striae, suppressed reactions to skin tests, thin fragile skin, thinning scalp hair, urticaria.
Endocrine
Decreased carbohydrate and glucose tolerance, development of cushingoid state, glucosuria, hirsutism, hypertrichosis, increased requirements for insulin or oral hypoglycemic adrenocortical and pituitary unresponsiveness (particularly in times of stress, as in trauma, surgery, or illness), suppression of growth in pediatric patients.
Fluid and Electrolyte Disturbances
Congestive heart failure in susceptible patients, fluid retention, hypokalemic alkalosis, potassium loss, sodium retention.
Gastrointestinal
Abdominal distention, bowel/bladder dysfunction (after intrathecal administration), elevation in serum liver enzyme levels (usually reversible upon discontinuation), hepatomegaly, increased appetite, nausea, pancreatitis, peptic ulcer with possible perforation and hemorrhage, perforation of the small and large intestine (particularly in patients with inflammatory bowel disease), ulcerative esophagitis.
Metabolic
Negative nitrogen balance due to protein catabolism.
Musculoskeletal
Aseptic necrosis of femoral and humeral heads, calcinosis (following intra-articular or intralesional use), Charcot-like arthropathy, loss of muscle mass, muscle weakness, osteoporosis, pathologic fracture of long bones, postinjection flare (following intra-articular use), steroid myopathy, tendon rupture, vertebral compression fractures.
Neurologic/Psychiatric
Convulsions, depression, emotional instability, euphoria, headache, increased intracranial pressure with papilledema (pseudotumor cerebri) usually following discontinuation of treatment, insomnia, mood swings, neuritis, neuropathy, paresthesia, personality changes, psychic disorders, vertigo. Arachnoiditis, meningitis, paraparesis/paraplegia, and sensory disturbances have occurred after intrathecal administration (see WARNINGS, Neurologicsection).
Ophthalmic
Exophthalmos, glaucoma, increased intraocular pressure, posterior subcapsular cataracts, rare instances of blindness associated with periocular injections, vision blurred.
Other
Abnormal fat deposits, decreased resistance to infection, hiccups, increased or decreased motility and number of spermatozoa, malaise, moon face, weight gain.
Overdosage
Treatment of acute overdose is by supportive and symptomatic therapy. For chronic overdosage in the face of severe disease requiring continuous steroid therapy, the dosage of the corticosteroid may be reduced only temporarily, or alternate day treatment may be introduced.
Dosage And Administration
Benzyl alcohol as a preservative has been associated with a fatal “Gasping Syndrome” in premature infants and infants of low birth weight. Solutions used for further dilution of this product should be preservative-free when used in the neonate, especially the premature infant. The initial dosage of parenterally administered Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension may vary from 0.25 to 9 mg per day depending on the specific disease entity being treated. However, in certain overwhelming, acute, life-threatening situations, administrations in dosages exceeding the usual dosages may be justified and may be in multiples of the oral dosages.
It Should Be Emphasized That Dosage Requirements Are Variable and Must Be Individualized on the Basis of the Disease Under Treatment and the Response of the Patient.After a favorable response is noted, the proper maintenance dosage should be determined by decreasing the initial drug dosage in small decrements at appropriate time intervals until the lowest dosage which will maintain an adequate clinical response is reached. Situations which may make dosage adjustments necessary are changes in clinical status secondary to remissions or exacerbations in the disease process, the patient’s individual drug responsiveness, and the effect of patient exposure to stressful situations not directly related to the disease entity under treatment. In this latter situation it may be necessary to increase the dosage of the corticosteroid for a period of time consistent with the patient’s condition. If after long-term therapy the drug is to be stopped, it is recommended that it be withdrawn gradually rather than abruptly.
In the treatment of acute exacerbations of multiple sclerosis, daily doses of 30 mg of betamethasone for a week followed by 12 mg every other day for 1 month are recommended (see PRECAUTIONS, Neuro-psychiatricsection).
In pediatric patients, the initial dose of betamethasone may vary depending on the specific disease entity being treated. The range of initial doses is 0.02 to 0.3 mg/kg/day in three or four divided doses (0.6 to 9 mg/m 2bsa/day).
For the purpose of comparison, the following is the equivalent milligram dosage of the various glucocorticoids: Cortisone, 25 Triamcinolone, 4 Hydrocortisone, 20 Paramethasone, 2 Prednisolone, 5 Betamethasone, 0.75 Prednisone, 5 Dexamethasone, 0.75 Methylprednisolone, 4
These dose relationships apply only to oral or intravenous administration of these compounds. When these substances or their derivatives are injected intramuscularly or into joint spaces, their relative properties may be greatly altered.
If coadministration of a local anesthetic is desired, Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension may be mixed with 1% or 2% lidocaine hydrochloride, using the formulations which do not contain parabens. Similar local anesthetics may also be used. Diluents containing methylparaben, propylparaben, phenol, etc., should be avoided, since these compounds may cause flocculation of the steroid. The required dose of Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension is first withdrawn from the vial into the syringe. The local anesthetic is then drawn in, and the syringe shaken briefly. Do not inject local anesthetics into the vial of Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension.
Bursitis, Tenosynovitis, Peritendinitis
In acute subdeltoid, subacromial, olecranon, and prepatellar bursitis, one intrabursal injection of 1 mL Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension can relieve pain and restore full range of movement. Several intrabursal injections of corticosteroids are usually required in recurrent acute bursitis and in acute exacerbations of chronic bursitis. Partial relief of pain and some increase in mobility can be expected in both conditions after one or two injections. Chronic bursitis may be treated with reduced dosage once the acute condition is controlled. In tenosynovitis and tendinitis, three or four local injections at intervals of 1 to 2 weeks between injections are given in most cases. Injections should be made into the affected tendon sheaths rather than into the tendons themselves. In ganglions of joint capsules and tendon sheaths, injection of 0.5 mL directly into the ganglion cysts has produced marked reduction in the size of the lesions.
Rheumatoid Arthritis and Osteoarthritis
Following intra-articular administration of 0.5 to 2 mL of Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension, relief of pain, soreness, and stiffness may be experienced. Duration of relief varies widely in both diseases. Intra-articular Injection of Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension is well tolerated in joints and periarticular tissues. There is virtually no pain on injection, and the “secondary flare” that sometimes occurs a few hours after intra-articular injection of corticosteroids has not been reported with Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension. Using sterile technique, a 20- to 24-gauge needle on an empty syringe is inserted into the synovial cavity and a few drops of synovial fluid are withdrawn to confirm that the needle is in the joint. The aspirating syringe is replaced by a syringe containing Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension and injection is then made into the joint.
Recommended Doses for Intra-articular Injection Size of Joint Location Dose (mL) Very Large Hip 1 - 2 Large Knee, ankle, shoulder 1 Medium Elbow, wrist 0.5 - 1 Small (metacarpophalangeal, interphalangeal) (sternoclavicular) Hand, chest 0.25 to 0.5
A portion of the administered dose of Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension is absorbed systemically following intra-articular injection. In patients being treated concomitantly with oral or parenteral corticosteroids, especially those receiving large doses, the systemic absorption of the drug should be considered in determining intra-articular dosage.
Dermatologic Conditions
In intralesional treatment, 0.2 mL/cm 2of Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension is injected intradermally (not subcutaneously) using a tuberculin syringe with a 25-gauge, 1/2-inch needle. Care should be taken to deposit a uniform depot of medication intradermally. A total of no more than 1 mL at weekly intervals is recommended.
Disorders of the Foot
A tuberculin syringe with a 25-gauge, 3/4-inch needle is suitable for most injections into the foot. The following doses are recommended at intervals of 3 days to a week.
Diagnosis Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension Dose (mL)
Bursitis under heloma durum or heloma molle
0.25 to 0.5 under calcaneal spur 0.5 over hallux rigidus or digiti quinti varus 0.5 Tenosynovitis, periostitis of cuboid 0.5 Acute gouty arthritis 0.5 to 1
How Supplied
NDC 0517-0720-01:
Betamethasone Sodium Phosphate and Betamethasone Acetate Injectable Suspension, 5 mL multiple dose vial; box of one. Inactive ingredients per mL: 7.1 mg dibasic sodium phosphate anhydrous; 3.4 mg monobasic sodium phosphate monohydrate; 0.1 mg edetate disodium; and 0.2 mg benzalkonium chloride as preservative.
SHAKE WELL BEFORE USING.
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from light.
Rx only
AMERICAN REGENT, INC. SHIRLEY, NY 11967
Revised July 2018
Lidocaine HCl Injection, USP
For Infiltration and Nerve Block Including Caudal and Epidural Use.
Preservative-Free
Rx only
Description
Lidocaine hydrochloride injection, USP is sterile, nonpyrogenic, aqueous solution that contains a local anesthetic agent and is administered parenterally by injection. See INDICATIONS AND USAGE section for specific uses. Lidocaine hydrochloride injection, USP contains lidocaine hydrochloride, which is chemically designated as acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-, monohydrochloride and has the molecular weight 270.8. Lidocaine hydrochloride (C 14H 22N 2O • HCl) has the following structural formula:
Lidocaine hydrochloride injection, USP is a sterile, nonpyrogenic, isotonic solution containing sodium chloride. The pH of the solution is adjusted to approximately 6.5 (5.0 to 7.0) with sodium hydroxide and/or hydrochloric acid.
Clinical Pharmacology
Mechanism of Action
Lidocaine hydrochloride stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses thereby effecting local anesthetic action.
Hemodynamics
Excessive blood levels may cause changes in cardiac output, total peripheral resistance, and mean arterial pressure. With central neural blockade these changes may be attributable to block of autonomic fibers, a direct depressant effect of the local anesthetic agent on various components of the cardiovascular system, and/or the beta-adrenergic receptor stimulating action of epinephrine when present. The net effect is normally a modest hypotension when the recommended dosages are not exceeded.
Pharmacokinetics and Metabolism
Information derived from diverse formulations, concentrations and usages reveals that lidocaine hydrochloride is completely absorbed following parenteral administration, its rate of absorption depending, for example, upon various factors such as the site of administration and the presence or absence of a vasoconstrictor agent. Except for intravascular administration, the highest blood levels are obtained following intercostal nerve block and the lowest after subcutaneous administration. The plasma binding of lidocaine hydrochloride is dependent on drug concentration, and the fraction bound decreases with increasing concentration. At concentrations of 1 to 4 mcg of free base per mL 60 to 80 percent of lidocaine hydrochloride is protein bound. Binding is also dependent on the plasma concentration of the alpha-1-acid glycoprotein. Lidocaine hydrochloride crosses the blood-brain and placental barriers, presumably by passive diffusion. Lidocaine hydrochloride is metabolized rapidly by the liver, and metabolites and unchanged drug are excreted by the kidneys. Biotransformation includes oxidative N-dealkylation, ring hydroxylation, cleavage of the amide linkage, and conjugation. N-dealkylation, a major pathway of biotransformation, yields the metabolites monoethylglycinexylidide and glycinexylidide. The pharmacological/toxicological actions of these metabolites are similar to, but less potent than, those of lidocaine hydrochloride. Approximately 90% of lidocaine hydrochloride administered is excreted in the form of various metabolites, and less than 10% is excreted unchanged. The primary metabolite in urine is a conjugate of 4-hydroxy-2,6-dimethylaniline. The elimination half-life of lidocaine hydrochloride following an intravenous bolus injection is typically 1.5 to 2 hours. Because of the rapid rate at which lidocaine hydrochloride is metabolized, any condition that affects liver function may alter lidocaine hydrochloride kinetics. The half-life may be prolonged two-fold or more in patients with liver dysfunction. Renal dysfunction does not affect lidocaine hydrochloride kinetics but may increase the accumulation of metabolites. Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine hydrochloride required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6 mcg free base per mL. In the rhesus monkey arterial blood levels of 18 to 21 mcg/mL have been shown to be threshold for convulsive activity.
Indications And Usage
Lidocaine hydrochloride injection is indicated for production of local or regional anesthesia by infiltration techniques such as percutaneous injection and intravenous regional anesthesia by peripheral nerve block techniques such as brachial plexus and intercostal and by central neural techniques such as lumbar and caudal epidural blocks, when the accepted procedures for these techniques as described in standard textbooks are observed.
Contraindications
Lidocaine hydrochloride is contraindicated in patients with a known history of hypersensitivity to local anesthetics of the amide type.
Warnings
LIDOCAINE HYDROCHLORIDE INJECTION FOR INFILTRATION AND NERVE BLOCK SHOULD BE EMPLOYED ONLY BY CLINICIANS WHO ARE WELL VERSED IN DIAGNOSIS AND MANAGEMENT OF DOSE-RELATED TOXICITY AND OTHER ACUTE EMERGENCIES THAT MIGHT ARISE FROM THE BLOCK TO BE EMPLOYED AND THEN ONLY AFTER ENSURING THE IMMEDIATEAVAILABILITY OF OXYGEN, OTHER RESUSCITATIVE DRUGS, CARDIOPULMONARY EQUIPMENT AND THE PERSONNEL NEEDED FOR PROPER MANAGEMENT OF TOXIC REACTIONS AND RELATED EMERGENCIES (see also ADVERSE REACTIONS and PRECAUTIONS ). DELAY IN PROPER MANAGEMENT OF DOSE-RELATED TOXICITY, UNDERVENTILATION FROM ANY CAUSE AND/OR ALTERED SENSITIVITY MAY LEAD TO THE DEVELOPMENT OF ACIDOSIS, CARDIAC ARREST AND, POSSIBLY, DEATH. Methemoglobinemia Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition. If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended. Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure, and are characterized by a cyanotic skin discoloration and/or abnormal coloration of the blood. Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue lidocaine hydrochloride and any other oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen. Intra-articular infusions of local anesthetics following arthroscopic and other surgical procedures is an unapproved use, and there have been post-marketing reports of chondrolysis in patients receiving such infusions. The majority of reported cases of chondrolysis have involved the shoulder joint; cases of gleno-humeral chondrolysis have been described in pediatric and adult patients following intra-articular infusions of local anesthetics with and without epinephrine for periods of 48 to 72 hours. There is insufficient information to determine whether shorter infusion periods are not associated with these findings. The time of onset of symptoms, such as joint pain, stiffness and loss of motion can be variable, but may begin as early as the 2 ndmonth after surgery. Currently, there is no effective treatment for chondrolysis; patients who experienced chondrolysis have required additional diagnostic and therapeutic procedures and some required arthroplasty or shoulder replacement. To avoid intravascular injection, aspiration should be performed before the local anesthetic solution is injected. The needle must be repositioned until no return of blood can be elicited by aspiration. Note, however, that the absence of blood in the syringe does not guarantee that intravascular injection has been avoided. Anaphylactic reactions may occur following administration of lidocaine hydrochloride (see ADVERSE REACTIONS ). In the case of severe reaction, discontinue the use of the drug.
Precautions
General
The safety and effectiveness of lidocaine hydrochloride depend on proper dosage, correct technique, adequate precautions, and readiness for emergencies. Standard textbooks should be consulted for specific techniques and precautions for various regional anesthetic procedures. Resuscitative equipment, oxygen, and other resuscitative drugs should be available for immediate use (see WARNINGS and ADVERSE REACTIONS ). The lowest dosage that results in effective anesthesia should be used to avoid high plasma levels and serious adverse effects. Syringe aspirations should also be performed before and during each supplemental injection when using indwelling catheter techniques. During the administration of epidural anesthesia, it is recommended that a test dose be administered initially and that the patient be monitored for central nervous system toxicity and cardiovascular toxicity, as well as for signs of unintended intrathecal administration, before proceeding. When clinical conditions permit, consideration should be given to employing local anesthetic solutions that contain epinephrine for the test dose because circulatory changes compatible with epinephrine may also serve as a warning sign of unintended intravascular injection. An intravascular injection is still possible even if aspirations for blood are negative. Repeated doses of lidocaine hydrochloride may cause significant increases in blood levels with each repeated dose because of slow accumulation of the drug or its metabolites. Tolerance to elevated blood levels varies with the status of the patient. Debilitated, elderly patients, acutely ill patients, and children should be given reduced doses commensurate with their age and physical condition. Lidocaine hydrochloride should also be used with caution in patients with severe shock or heart block. Lumbar and caudal epidural anesthesia should be used with extreme caution in persons with the following conditions: existing neurological disease, spinal deformities, septicemia, and severe hypertension. Local anesthetic solutions containing a vasoconstrictor should be used cautiously and in carefully circumscribed quantities in areas of the body supplied by end arteries or having otherwise compromised blood supply. Patients with peripheral vascular disease and those with hypertensive vascular disease may exhibit exaggerated vasoconstrictor response. Ischemic injury or necrosis may result. Preparations containing a vasoconstrictor should be used with caution in patients during or following the administration of potent general anesthetic agents, since cardiac arrhythmias may occur under such conditions. Careful and constant monitoring of cardiovascular and respiratory (adequacy of ventilation) vital signs and the patient’s state of consciousness should be accomplished after each local anesthetic injection. It should be kept in mind at such times that restlessness, anxiety, tinnitus, dizziness, blurred vision, tremors, depression or drowsiness may be early warning signs of central nervous system toxicity. Since amide-type local anesthetics are metabolized by the liver, lidocaine hydrochloride injection should be used with caution in patients with hepatic disease. Patients with severe hepatic disease, because of their inability to metabolize local anesthetics normally, are at greater risk of developing toxic plasma concentrations. Lidocaine hydrochloride injection should also be used with caution in patients with impaired cardiovascular function since they may be less able to compensate for functional changes associated with the prolongation of A-V conduction produced by these drugs. Many drugs used during the conduct of anesthesia are considered potential triggering agents for familial malignant hyperthermia. Since it is not known whether amide-type local anesthetics may trigger this reaction and since the need for supplemental general anesthesia cannot be predicted in advance, it is suggested that a standard protocol for the management of malignant hyperthermia should be available. Early unexplained signs of tachycardia, tachypnea, labile blood pressure and metabolic acidosis may precede temperature elevation. Successful outcome is dependent on early diagnosis, prompt discontinuance of the suspect triggering agent(s) and institution of treatment, including oxygen therapy, indicated supportive measures and dantrolene (consult dantrolene sodium intravenous package insert before using). Proper tourniquet technique, as described in publications and standard textbooks, is essential in the performance of intravenous regional anesthesia. Solutions containing epinephrine or other vasoconstrictors should not be used for this technique. Lidocaine hydrochloride should be used with caution in persons with known drug sensitivities. Patients allergic to para-aminobenzoic acid derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross-sensitivity to lidocaine hydrochloride.
Use in the Head and Neck Area
Small doses of local anesthetics injected into the head and neck area, including retrobulbar, dental and stellate ganglion blocks, may produce adverse reactions similar to systemic toxicity seen with unintentional intravascular injections of larger doses. Confusion, convulsions, respiratory depression and/or respiratory arrest, and cardiovascular stimulation or depression have been reported. These reactions may be due to intra-arterial injection of the local anesthetic with retrograde flow to the cerebral circulation. Patients receiving these blocks should have their circulation and respiration monitored and be constantly observed. Resuscitative equipment and personnel for treating adverse reactions should be immediately available. Dosage recommendations should not be exceeded (see DOSAGE AND ADMINISTRATION ).
Information for Patients
When appropriate, patients should be informed in advance that they may experience temporary loss of sensation and motor activity, usually in the lower half of the body, following proper administration of epidural anesthesia. Inform patients that use of local anesthetics may cause methemoglobinemia, a serious condition that must be treated promptly. Advise patients or caregivers to seek immediate medical attention if they or someone in their care experience the following signs or symptoms: pale, gray, or blue colored skin (cyanosis); headache; rapid heart rate; shortness of breath; lightheadedness; or fatigue.
Clinically Significant Drug Interactions
The administration of local anesthetic solutions containing epinephrine or norepinephrine to patients receiving monoamine oxidase inhibitors or tricyclic antidepressants may produce severe, prolonged hypertension. Phenothiazines and butyrophenones may reduce or reverse the pressor effect of epinephrine. Concurrent use of these agents should generally be avoided. In situations when concurrent therapy is necessary, careful patient monitoring is essential. Concurrent administration of vasopressor drugs (for the treatment of hypotension related to obstetric blocks) and ergot-type oxytocic drugs may cause severe, persistent hypertension or cerebrovascular accidents.
Drug/Laboratory Test Interactions
The intramuscular injection of lidocaine hydrochloride may result in an increase in creatine phosphokinase levels. Thus, the use of this enzyme determination, without isoenzyme separation, as a diagnostic test for the presence of acute myocardial infarction may be compromised by the intramuscular injection of lidocaine hydrochloride. Patients who are administered local anesthetics are at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
Examples of Drugs Associated with Methemoglobinemia: Class Examples Nitrates/Nitrites nitric oxide, nitroglycerin, nitroprusside, nitrous oxide Local anesthetics articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine Antineoplastic agents cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase Antibiotics dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides Antimalarials chloroquine, primaquine Anticonvulsants Phenobarbital, phenytoin, sodium valproate Other drugs acetaminophen, metoclopramide, quinine, sulfasalazine Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies of lidocaine hydrochloride in animals to evaluate the carcinogenic and mutagenic potential or the effect on fertility have not been conducted.
Pregnancy
Teratogenic Effects
Reproduction studies have been performed in rats at doses up to 6.6 times the human dose and have revealed no evidence of harm to the fetus caused by lidocaine hydrochloride. There are, however, no adequate and well-controlled studies in pregnant women. Animal reproduction studies are not always predictive of human response. General consideration should be given to this fact before administering lidocaine hydrochloride to women of childbearing potential, especially during early pregnancy when maximum organogenesis takes place.
Labor and Delivery
Local anesthetics rapidly cross the placenta and when used for epidural, paracervical, pudendal or caudal block anesthesia, can cause varying degrees of maternal, fetal and neonatal toxicity (see CLINICAL PHARMACOLOGY , Pharmacokinetics and Metabolism ). The potential for toxicity depends upon the procedure performed, the type and amount of drug used, and the technique of drug administration. Adverse reactions in the parturient, fetus and neonate involve alterations of the central nervous system, peripheral vascular tone and cardiac function. Maternal hypotension has resulted from regional anesthesia. Local anesthetics produce vasodilation by blocking sympathetic nerves. Elevating the patient’s legs and positioning her on her left side will help prevent decreases in blood pressure. The fetal heart rate also should be monitored continuously, and electronic fetal monitoring is highly advisable. Epidural, spinal, paracervical, or pudendal anesthesia may alter the forces of parturition through changes in uterine contractility or maternal expulsive efforts. In one study, paracervical block anesthesia was associated with a decrease in the mean duration of first stage labor and facilitation of cervical dilation. However, spinal and epidural anesthesia have also been reported to prolong the second stage of labor by removing the parturient’s reflex urge to bear down or by interfering with motor function. The use of obstetrical anesthesia may increase the need for forceps assistance. The use of some local anesthetic drug products during labor and delivery may be followed by diminished muscle strength and tone for the first day or two of life. The long-term significance of these observations is unknown. Fetal bradycardia may occur in 20 to 30 percent of patients receiving paracervical nerve block anesthesia with the amide-type local anesthetics and may be associated with fetal acidosis. Fetal heart rate should always be monitored during paracervical anesthesia. The physician should weigh the possible advantages against risks when considering a paracervical block in prematurity, toxemia of pregnancy, and fetal distress. Careful adherence to recommended dosage is of the utmost importance in obstetrical paracervical block. Failure to achieve adequate analgesia with recommended doses should arouse suspicion of intravascular or fetal intracranial injection. Cases compatible with unintended fetal intracranial injection of local anesthetic solution have been reported following intended paracervical or pudendal block or both. Babies so affected present with unexplained neonatal depression at birth, which correlates with high local anesthetic serum levels, and often manifest seizures within six hours. Prompt use of supportive measures combined with forced urinary excretion of the local anesthetic has been used successfully to manage this complication. Case reports of maternal convulsions and cardiovascular collapse following use of some local anesthetics for paracervical block in early pregnancy (as anesthesia for elective abortion) suggest that systemic absorption under these circumstances may be rapid. The recommended maximum dose of each drug should not be exceeded. Injection should be made slowly and with frequent aspiration. Allow a 5-minute interval between sides.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when lidocaine hydrochloride is administered to a nursing woman.
Pediatric Use
Dosages in children should be reduced, commensurate with age, body weight and physical condition, see DOSAGE AND ADMINISTRATION .
Adverse Reactions
Systemic
Adverse experiences following the administration of lidocaine hydrochloride are similar in nature to those observed with other amide local anesthetic agents. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage, rapid absorption or inadvertent intravascular injection, or may result from a hypersensitivity, idiosyncrasy or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported:
CNS manifestations are excitatory and/or depressant and may be characterized by lightheadedness, nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred or double vision, vomiting, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, respiratory depression and arrest. The excitatory manifestations may be very brief or may not occur at all, in which case the first manifestation of toxicity may be drowsiness merging into unconsciousness and respiratory arrest.
Drowsiness following the administration of lidocaine hydrochloride is usually an early sign of a high blood level of the drug and may occur as a consequence of rapid absorption.
Cardiovascular manifestations are usually depressant and are characterized by bradycardia, hypotension, and cardiovascular collapse, which may lead to cardiac arrest.
Allergic reactions are characterized by cutaneous lesions, urticaria, edema or anaphylactoid reactions. Allergic reactions may occur as a result of sensitivity to local anesthetic agents. Allergic reactions, including anaphylactic reactions, may occur as a result of sensitivity to lidocaine, but are infrequent. If allergic reactions do occur, they should be managed by conventional means. The detection of sensitivity by skin testing is of doubtful value. There have been no reports of cross sensitivity between lidocaine hydrochloride and procainamide or between lidocaine hydrochloride and quinidine.
The incidences of adverse reactions associated with the use of local anesthetics may be related to the total dose of local anesthetic administered and are also dependent upon the particular drug used, the route of administration and the physical status of the patient. In a prospective review of 10,440 patients who received lidocaine hydrochloride for spinal anesthesia, the incidences of adverse reactions were reported to be about 3 percent each for positional headaches, hypotension and backache; 2 percent for shivering; and less than 1 percent each for peripheral nerve symptoms, nausea, respiratory inadequacy and double vision. Many of these observations may be related to local anesthetic techniques, with or without a contribution from the local anesthetic. In the practice of caudal or lumbar epidural block, occasional unintentional penetration of the subarachnoid space by the catheter may occur. Subsequent adverse effects may depend partially on the amount of drug administered subdurally. These may include spinal block of varying magnitude (including total spinal block), hypotension secondary to spinal block, loss of bladder and bowel control, and loss of perineal sensation and sexual function. Persistent motor, sensory and/or autonomic (sphincter control) deficit of some lower spinal segments with slow recovery (several months) or incomplete recovery have been reported in rare instances when caudal or lumbar epidural block has been attempted. Backache and headache have also been noted following use of these anesthetic procedures. There have been reported cases of permanent injury to extraocular muscles requiring surgical repair following retrobulbar administration.
Methemoglobinemia.
Overdosage
Acute emergencies from local anesthetics are generally related to high plasma levels encountered during therapeutic use of local anesthetics or to unintended subarachnoid injection of local anesthetic solution (see ADVERSE REACTIONS , WARNINGS , and PRECAUTIONS ).
Management of Local Anesthetic Emergencies
The first consideration is prevention, best accomplished by careful and constant monitoring of cardiovascular and respiratory vital signs and the patient’s state of consciousness after each local anesthetic injection. At the first sign of change, oxygen should be administered. The first step in the management of convulsions, as well as underventilation or apnea due to unintended subarachnoid injection of drug solution, consists of immediate attention to the maintenance of a patent airway and assisted or controlled ventilation with oxygen and a delivery system capable of permitting immediate positive airway pressure by mask. Immediately after the institution of these ventilatory measures, the adequacy of the circulation should be evaluated, keeping in mind that drugs used to treat convulsions sometimes depress the circulation when administered intravenously. Should convulsions persist despite adequate respiratory support, and if the status of the circulation permits, small increments of an ultra-short acting barbiturate (such as thiopental or thiamylal) or a benzodiazepine (such as diazepam) may be administered intravenously. The clinician should be familiar, prior to the use of local anesthetics, with these anticonvulsant drugs. Supportive treatment of circulatory depression may require administration of intravenous fluids and, when appropriate, a vasopressor as directed by the clinical situation (e.g., ephedrine). If not treated immediately, both convulsions and cardiovascular depression can result in hypoxia, acidosis, bradycardia, arrhythmias and cardiac arrest. Underventilation or apnea due to unintentional subarachnoid injection of local anesthetic solution may produce these same signs and also lead to cardiac arrest if ventilatory support is not instituted. If cardiac arrest should occur, standard cardiopulmonary resuscitative measures should be instituted. Endotracheal intubation, employing drugs and techniques familiar to the clinician, may be indicated, after initial administration of oxygen by mask, if difficulty is encountered in the maintenance of a patent airway or if prolonged ventilatory support (assisted or controlled) is indicated. Dialysis is of negligible value in the treatment of acute overdosage with lidocaine hydrochloride. The oral LD 50of lidocaine hydrochloride in non-fasted female rats is 459 (346 to 773) mg/kg (as the salt) and 214 (159 to 324) mg/kg (as the salt) in fasted female rats.
Dosage And Administration
Table 1 (Recommended Dosages) summarizes the recommended volumes and concentrations of lidocaine hydrochloride injection for various types of anesthetic procedures. The dosages suggested in this table are for normal healthy adults and refer to the use of epinephrine-free solutions. When larger volumes are required, only solutions containing epinephrine should be used except in those cases where vasopressor drugs may be contraindicated. There have been adverse event reports of chondrolysis in patients receiving intra-articular infusions of local anesthetics following arthroscopic and other surgical procedures. Lidocaine hydrochloride injection is not approved for this use (see WARNINGS and DOSAGE AND ADMINISTRATION ). These recommended doses serve only as a guide to the amount of anesthetic required for most routine procedures. The actual volumes and concentrations to be used depend on a number of factors such as type and extent of surgical procedure, depth of anesthesia and degree of muscular relaxation required, duration of anesthesia required, and the physical condition of the patient. In all cases the lowest concentration and smallest dose that will produce the desired result should be given. Dosages should be reduced for children and for the elderly and debilitated patients and patients with cardiac and/or liver disease. The onset of anesthesia, the duration of anesthesia and the degree of muscular relaxation are proportional to the volume and concentration (i.e., total dose) of local anesthetic used. Thus, an increase in volume and concentration of lidocaine hydrochloride injection will decrease the onset of anesthesia, prolong the duration of anesthesia, provide a greater degree of muscular relaxation and increase the segmental spread of anesthesia. However, increasing the volume and concentration of lidocaine hydrochloride injection may result in a more profound fall in blood pressure when used in epidural anesthesia. Although the incidence of side effects with lidocaine hydrochloride is quite low, caution should be exercised when employing large volumes and concentrations, since the incidence of side effects is directly proportional to the total dose of local anesthetic agent injected.
Epidural Anesthesia
For epidural anesthesia the following dosage form of lidocaine hydrochloride injection is recommended: 1% without epinephrine 30 mL single dose vials Although this solution is intended specifically for epidural anesthesia, it may also be used for infiltration and peripheral nerve block, provided it is employed as a single dose unit. This solution contains no bacteriostatic agent. In epidural anesthesia, the dosage varies with the number of dermatomes to be anesthetized (generally 2 to 3 mL of the indicated concentration per dermatome).
Caudal and Lumbar Epidural Block
As a precaution against the adverse experience sometimes observed following unintentional penetration of the subarachnoid space, a test dose such as 2 to 3 mL of 1.5% lidocaine hydrochloride should be administered at least 5 minutes prior to injecting the total volume required for a lumbar or caudal epidural block. The test dose should be repeated if the patient is moved in a manner that may have displaced the catheter. Epinephrine, if contained in the test dose (10 to 15 mcg have been suggested), may serve as a warning of unintentional intravascular injection. If injected into a blood vessel, this amount of epinephrine is likely to produce a transient “epinephrine response” within 45 seconds, consisting of an increase in heart rate and systolic blood pressure, circumoral pallor, palpitations and nervousness in the unsedated patient. The sedated patient may exhibit only a pulse rate increase of 20 or more beats per minute for 15 or more seconds. Patients on beta blockers may not manifest changes in heart rate, but blood pressure monitoring can detect an evanescent rise in systolic blood pressure. Adequate time should be allowed for onset of anesthesia after administration of each test dose. The rapid injection of a large volume of lidocaine hydrochloride injection through the catheter should be avoided, and, when feasible, fractional doses should be administered. In the event of the known injection of a large volume of local anesthetic solution into the subarachnoid space, after suitable resuscitation and if the catheter is in place, consider attempting the recovery of drug by draining a moderate amount of cerebrospinal fluid (such as 10 mL) through the epidural catheter.
Maximum Recommended Dosages
Adults
For normal healthy adults, the maximum individual dose should not exceed 4.5 mg/kg (2 mg/lb) of body weight, and in general it is recom mended that the max i m um t otal dose does not exceed 300 mg. For continuous epidural or caudal anesthesia, the maximum recommended dosage should not be administered at intervals of less than 90 minutes. When continuous lumbar or caudal epidural anesthesia is used for non-obstetrical procedures, more drug may be administered if required to produce adequate anesthesia. The maximum recommended dose per 90 minute period of lidocaine hydrochloride for paracervical block in obstetrical patients and non-obstetrical patients is 200 mg total. One half of the total dose is usually administered to each side. Inject slowly, five minutes between sides (see also discussion of paracervical block in PRECAUTIONS ). For intravenous regional anesthesia, the dose administered should not exceed 4 mg/kg in adults.
Children
It is difficult to recommend a maximum dose of any drug for children, since this varies as a function of age and weight. For children over 3 years of age who have a normal lean body mass and normal body development, the maximum dose is determined by the child’s age and weight. For example, in a child of 5 years weighing 50 lbs the dose of lidocaine hydrochloride should not exceed 75 to 100 mg (1.5 to 2 mg/lb). The use of even more dilute solutions (i.e., 0.25 to 0.5%) and total dosages not to exceed 3 mg/kg (1.4 mg/lb) are recommended for induction of intravenous regional anesthesia in children. In order to guard against systemic toxicity, the lowest effective concentration and lowest effective dose should be used at all times. In some cases it will be necessary to dilute available concentrations with 0.9% sodium chloride injection in order to obtain the required final concentration. NOTE: Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever the solution and container permit. Do not use if solution is discolored or contains a precipitate.
Table 1: Recommended Dosages Procedure Lidocaine Hydrochloride Injection (without epinephrine) Conc (%) Vol (mL) Total Dose (mg) *Dose determined by number of dermatomes to be anesthetized (2 to 3 mL/dermatome). Infiltration Percutaneous 0.5 or 1 1 to 60 5 to 300 Intravenous regional 0.5 10 to 60 50 to 300 Peripheral Nerve Blocks, e.g., Brachial 1.5 15 to 20 225 to 300 Dental 2 1 to 5 20 to 100 Intercostal 1 3 30 Paravertebral 1 3 to 5 30 to 50 Pudendal (each side) 1 10 100 Paracervical Obstetrical analgesia (each side) 1 10 100 Sympathetic Nerve Blocks, e.g., Cervical (stellate ganglion) 1 5 50 Lumbar 1 5 to 10 50 to 100 Central Neural Blocks Epidural* Thoracic 1 20 to 30 200 to 300 Lumbar Analgesia 1 25 to 30 250 to 300 Anesthesia 1.5 15 to 20 225 to 300 2 10 to 15 200 to 300 Caudal Obstetrical analgesia 1 20 to 30 200 to 300 Surgical anesthesia 1.5 15 to 20 225 to 300
THE ABOVE SUGGESTED CONCENTRATIONS AND VOLUMES SERVE ONLY AS A GUIDE. OTHER VOLUMES AND CONCENTRATIONS MAY BE USED PROVIDED THE TOTAL MAXIMUM RECOMMENDED DOSE IS NOT EXCEEDED.
Sterilization, Storage And Technical Procedures
Disinfecting agents containing heavy metals, which cause release of respective ions (mercury, zinc, copper, etc.) should not be used for skin or mucous membrane disinfection as they have been related to incidents of swelling and edema.
How Supplied
Lidocaine Hydrochloride Injection, USP is supplied as follows: Lidocaine Hydrochloride Injection USP, 1% (10 mg/mL) 5 mL Single Dose Vials            NDC 55150-162-05 Sterile, Nonpyrogenic Discard unused portion Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] The vial stopper is not made with natural rubber latex. Distributed by: AuroMedics Pharma LLC
279 Princeton-Hightstown Rd. E. Windsor, NJ 08520 Manufactured by: Aurobindo Pharma Limited
Hyderabad - 500038 India Revised: February 2020
Povidine Iodine Swab
Otc - Active Ingredient Section
Active Ingredient                                                 Purpose
Povidone Iodine 10% w/v (9.85% w/w/)Â Â Â Â Â Â Â Â Â Â Â Â Â Â Antiseptic
Purpose:
Purpose:
- First aid antiseptic to help prevent skin infection in minor cuts, scrapes and burns.
- For preparation of the skin prior to surgery.
- Helps reduce bacteria that can potentially cause skin infections.
Warnings:
Section Text
- FOR EXTERNAL USE ONLY
Do Not Use:
- As a first aid antiseptic for more than 1 week.
- In the eyes.
- Over large areas of the body.
Ask A Doctor Before Use If You Have:
- Deep puncture wounds
- Animal bites
- Serious burns
Stop Use:
- If irritation and redness develop
- If condition persists for more than 72 hours, consult a physician.
Keep Out Of Reach Of Children
Keep out of reach of children.If swallowed, get medical help or contact a Poison Control Center.
Directions Povidone Iodine:
Tear at notch, remove applicator, use only once.
As a first aid antiseptic
- clean affected area
- apply 1 to 3 times daily
- may be covered with a sterile bandage, if bandaged let dry.
For preoperative patient skin preparation
- clean area
- apply to operative site prior to surgery using the applicator
Other Information:
Store at room temperature.
Avoid excessive heat
Indications & Usage Section
For use as an
- first aid antiseptic
- pre-operative skin preperation
Inactive Ingredients
Inactive ingredients: Citric acid, glycerin, polysorbate 80, sodium citrate USP, sodium phosphate dibasic, water
Isopropyl Alcohol 70% Prep Pads
Active Ingredient
Purpose
Antiseptic
Uses
For first aid to decrease germs in
- minor cuts
- scrapes
- burns
For preparation of the skin prior to injection
Warnings
For external use only
Flammable - keep away from fire or flame
Do not use
with electrocautery procedures
When using this product do not
- get into eyes
- apply over large areas of the body
- in case of deep or puncture wounds, animal bites or serious burns consult a doctor
Stop use and ask a doctor if
- condition persists or gets worse or lasts for more than 72 hours
- do not use longer than 1 week unless directed by a doctor
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- apply to skin as needed
- discard after single use
Other Information
Protect from freezing and avoid excessive heat
Inactive Ingredient
Water
Package Label.principal Display Panel
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC: 76420-793-01
RX Only
BL-Contrast™
Kit Contains
1 Betamethasone Sodium Phosphate and Betamethasone Acetate 6mg/mL (5mL)
2 Lidocaine HCl Injection, USP 1% Single Dose Vial (5mL)
1 Omnipaque™ (iohexol) Injection 240mg/mL Single Dose Bottle (10mL)
1 Povidone-Iodine Swabsticks (3 Swabs)
3 Isopropyl Alcohol 70% Prep Pads
1 Pair Nitrile Powder Free Sterile Gloves (M)
1 Drape
1 Adhesive Bandage
5 Non Sterile 4x4 Gauze
Needles and Syringes Not Included
1 Dose
Single Use Only
Distributed by:
Enovachem Pharmaceuticals
Torrance, CA 90501

DISCLAIMER:
"This tool does not provide medical advice, and is for informational and educational purposes only, and is not a substitute for professional medical advice, treatment or diagnosis. Call your doctor to receive medical advice. If you think you may have a medical emergency, please dial 911."
"Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. We may limit or otherwise restrict your access to the API in line with our Terms of Service."
"This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product."
PillSync may earn a commission via links on our site

