Tobramycin Dailymed
Generic: tobramycin is used for the treatment of Bone Diseases, Infectious Central Nervous System Infections Pseudomonas Infections Respiratory Tract Infections Skin Diseases, Infectious Staphylococcal Infections Urinary Tract Infections Eye Infections, Bacterial Sepsis Intraabdominal Infections
Go PRO for all pill images
Rx only
Description:
Tobramycin Ophthalmic Solution USP, 0.3% is a sterile topical ophthalmic antibiotic formulation prepared specifically for topical therapy of external ophthalmic infections.
Tobramycin is a water soluble aminoglycoside antibiotic active against a wide variety of gram-negative and gram-positive ophthalmic pathogens. The molecular formula is C18H37N5O9, the molecular weight is 467.52 and the structural formula is:
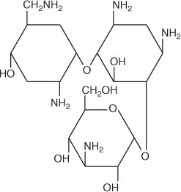
Chemical Name:
O-{3-amino-3-deoxy-α-D-gluco-pyranosyl-(1 → 4)}-O-{2,6-diamino-2,3,6-trideoxy-α-D-ribohexo-pyranosyl-(1 → 6)}-2- deoxystreptamine.
Each mL contains:
Active: Tobramycin 3 mg (0.3%). Inactives: Boric Acid, Sodium Chloride, Sodium Sulfate, Tyloxapol, Sodium Hydroxide and/or Sulfuric Acid to adjust pH (7.0 to 8.0), and Purified Water USP. Preservative: Benzalkonium Chloride 0.1 mg (0.01%).
Clinical Pharmacology:
In Vitro Data: In Vitro studies have demonstrated tobramycin is active against susceptible strains of the following microorganisms:
Staphylococci, including S. aureus and S. epidermidis (coagulase-positive and coagulase-negative), including penicillin-resistant strains.
Streptococci, including some of the Group A betahemolytic species, some nonhemolytic species, and some Streptococcus pneumoniae.
Pseudomonas aeruginosa, Escherichia coli, Klebsiella pneumoniae, Enterobacter aerogenes, Proteus mirabilis, Morganella morganii, most Proteus vulgaris strains, Haemophilus influenzae and H. aegyptius, Moraxella lacunata, Acinetobacter calcoaceticus and some Neisseria species.
Bacterial susceptibility studies demonstrate that in some cases, microorganisms resistant to gentamicin retain susceptibility to tobramycin.
Indications And Usage:
Tobramycin Ophthalmic Solution USP, 0.3% is a topical antibiotic indicated in the treatment of external infections of the eye and its adnexa caused by susceptible bacteria. Appropriate monitoring of bacterial response to topical antibiotic therapy should accompany the use of Tobramycin Ophthalmic Solution USP, 0.3%. Clinical studies have shown tobramycin to be safe and effective for use in pediatric patients.
Contraindications:
Tobramycin Ophthalmic Solution USP, 0.3% is contraindicated in patients with known hypersensitivity to any of its components.
Warnings:
FOR TOPICAL OPHTHALMIC USE ONLY. NOT FOR INJECTION INTO THE EYE. Sensitivity to topically applied aminoglycosides may occur in some patients. If a sensitivity reaction to Tobramycin Ophthalmic Solution USP, 0.3% occurs, discontinue use.
Precautions:
General:
As with other antibiotic preparations, prolonged use may result in overgrowth of nonsusceptible organisms, including fungi. If superinfection occurs, appropriate therapy should be initiated. Cross-sensitivity to other aminoglycoside antibiotics may occur; if hypersensitivity develops with this product, discontinue use and institute appropriate therapy. Patients should be advised not to wear contact lenses if they have signs and symptoms of bacterial conjunctivitis.
INFORMATION FOR PATIENTS SECTION
Information for Patients: Do not touch dropper tip to any surface, as this may contaminate the solution.
PREGNANCY SECTION
Pregnancy Category B: Reproduction studies in three types of animals at doses up to thirty-three times the normal human systemic dose have revealed no evidence of impaired fertility or harm to the fetus due to tobramycin. There are, however, no adequate and wellcontrolled studies in pregnant women. Because animal studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
NURSING MOTHERS SECTION
Nursing Mothers: Because of the potential for adverse reactions in nursing infants from tobramycin, a decision should be made whether to discontinue nursing the infant or discontinue the drug, taking into account the importance of the drug to the mother.
PEDIATRIC USE SECTION
Pediatric Use: Safety and effectiveness in pediatric patients below the age of two months has not been established.
GERIATRIC USE SECTION
Geriatric Use: No overall clinical differences in safety or effectiveness have been observed between the elderly and other adult patients.
Adverse Reactions:
The most frequent adverse reactions to tobramycin ophthalmic solution are hypersensitivity and localized ocular toxicity, including lid itching and swelling, and conjunctival erythema. These reactions occur in less than three of 100 patients treated with tobramycin. Similar reactions may occur with the topical use of other aminoglycoside antibiotics. Other adverse reactions have not been reported from tobramycin therapy; however, if topical ocular tobramycin is administered concomitantly with systemic aminoglycoside antibiotics, care should be taken to monitor the total serum concentration.
Overdosage:
Clinically apparent signs and symptoms of an overdose of tobramycin ophthalmic solution (punctate keratitis, erythema, increased lacrimation, edema and lid itching) may be similar to adverse reaction effects seen in some patients.
Dosage And Administration:
In mild to moderate disease, instill one or two drops into the affected eye(s) every four hours. In severe infections, instill two drops into the eye(s) hourly until improvement, following which treatment should be reduced prior to discontinuation.
How Supplied:
Tobramycin Ophthalmic Solution USP, 0.3% is supplied as a sterile solution in plastic dropper bottles in two sizes:
NDC 63187-902-02. . . . . . . . . . . . 2 mL
NDC 63187-902-05. . . . . . . . . . . . 5 mL
Storage:
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Keep container tightly closed.
Protect from excessive heat.
Akorn Manufactured by: Akorn, Inc. Lake Forest, IL 60045
Relabeled by: Proficient Rx LP Thousand Oaks, CA 91320
GTM00N Rev. 11/11
Package Label.principal Display Panel
Principal Display Panel Text for Container Label:
NDC 63187-902-05
Tobramycin
Ophthalmic
Solution, USP
0.3%
5 mL
Sterile
Rx only

DISCLAIMER:
"This tool does not provide medical advice, and is for informational and educational purposes only, and is not a substitute for professional medical advice, treatment or diagnosis. Call your doctor to receive medical advice. If you think you may have a medical emergency, please dial 911."
"Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. We may limit or otherwise restrict your access to the API in line with our Terms of Service."
"This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product."
PillSync may earn a commission via links on our site


