Zyvana Dailymed
Generic: zyvana
Go PRO for all pill images
Description
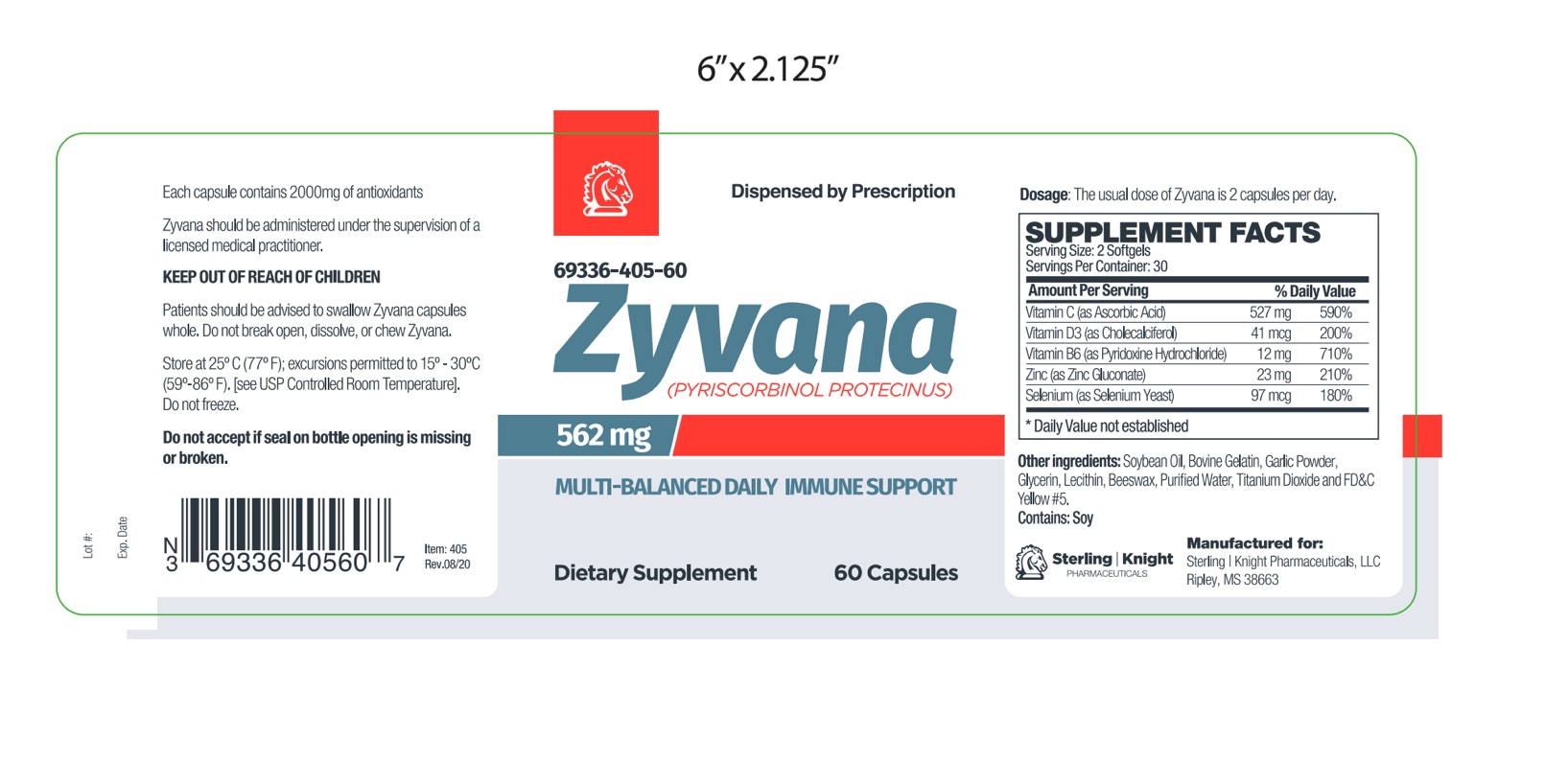
Zyvana is an orally administered prescription dietary supplement formulation.
Zyvana should be administered under the supervision of a licensed medical practitioner.

Other Ingredients: Soybean Oil, Gelatin (bovine), Garlic Powder, Glycerin, Lecithin, Beeswax, Purified Water, Titanium Dioxide, FD&C Yellow #5.
Indications And Usage
Zyvana is an orally administered prescription dietary supplement formulation for the management of suboptimal nutritional status in patients where advanced supplementation is required and nutritional supplementation in physiologically stressful conditions for maintenance of good health is needed.
Warnings And Precautions:
This product is contraindicated in patients with a known hypersensitivity to any of the ingredients.Zyvana should only be used under the direction and supervision of a licensed medical practitioner. Use with caution in patients that may have a medical condition, are pregnant, lactating, trying to conceive, under the age of 18, or taking medications.
Dosage And Administration
The usual dose of Zyvana is 2 capsules per day.The daily dose may be taken as a single dose of 2 capsules or 1 capsule twice daily.
How Supplied Section
Zyvana is supplied as yellow softgel capsule printed with 405 dispensed in white HDPE plastic bottles of 60ct. 69336-405-60
Storage
Store at controlled room temperature 15°-30°C (59°F-86°F). Keep in cool dry place. Call your doctor about side effects. You may report side effects to FDA at 1-800-FDA-1088. KEEP THIS OUT OF THE REACH OF CHILDREN.
Reserved For Professional Recommendation
All prescriptions using this dietary supplement product shall be pursuant to state statutes as applicable. This is not an Orange Book product. This product may be administered under a physician’s supervision. There are no implied or explicit claims on therapeutic equivalence.
Package Label.principal Display Panel
Manufactured for:
Sterling-Knight Pharmaceuticals, LLC
Ripley, MS 38663Â
Item 40560
Rev. 0920-1
DISCLAIMER:
"This tool does not provide medical advice, and is for informational and educational purposes only, and is not a substitute for professional medical advice, treatment or diagnosis. Call your doctor to receive medical advice. If you think you may have a medical emergency, please dial 911."
"Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. We may limit or otherwise restrict your access to the API in line with our Terms of Service."
"This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product."
PillSync may earn a commission via links on our site


