dextroamphetamine saccharate amphetamine aspartate dextroamphetamine sulfate and amphetamine sulfate tablet
INDICATIONS AND USAGE Dextroamphetamine saccharate, amphetamine aspartate, dextroamphetamine sulfate and amphetamine sulfate tablets are indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) and Narcolepsy. Attention Deficit Hyperactivity Disorder (ADHD) A diagnosis of Attention Deficit Hyperactivity Disorder (ADHD; DSM-IV ® ) implies the presence of hyperactive-impulsive or inattentive symptoms that caused impairment and were present before age 7 years. The symptoms must cause clinically significant impairment, e.g., in social, academic, or occupational functioning, and be present in two or more settings, e.g., school (or work) and at home. The symptoms must not be better accounted for by another mental disorder. For the Inattentive Type, at least six of the following symptoms must have persisted for at least 6 months: lack of attention to details/careless mistakes; lack of sustained attention; poor listener; failure to follow through on tasks; poor organization; avoids tasks requiring sustained mental effort; loses things; easily distracted; forgetful. For the Hyperactive-Impulsive Type, at least six of the following symptoms must have persisted for at least 6 months: fidgeting/squirming; leaving seat; inappropriate running/climbing; difficulty with quiet activities; "on the go;" excessive talking; blurting answers; can't wait turn; intrusive. The Combined Type requires both inattentive and hyperactive-impulsive criteria to be met.
Epic Pharma LLC
Related Pills
DISCLAIMER:
"This tool does not provide medical advice, and is for informational and educational purposes only, and is not a substitute for professional medical advice, treatment or diagnosis. Call your doctor to receive medical advice. If you think you may have a medical emergency, please dial 911."
"Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. We may limit or otherwise restrict your access to the API in line with our Terms of Service."
"This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product."
PillSync may earn a commission via links on our site










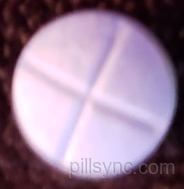





HOW SUPPLIED
DEXTROAMPHETAMINE SACCHARATE, AMPHETAMINE ASPARTATE, DEXTROAMPHETAMINE SULFATE AND AMPHETAMINE SULFATE tablets (Mixed Salts of a Single Entity Amphetamine Product) is supplied as follows: 5 mg: blue, round flat-faced beveled edge tablets, debossed "є" above "339" on one side and quadrisect on the other side. They are available in bottles of 100 tablets (NDC 42806-339-01). 10 mg: blue, round biconvex tablets, debossed “ є ” above “ 341 ” on one side and quadrisect on the other side. They are available in bottles of 100 tablets (NDC 42806-341-01) 15 mg: pink, oval-shaped tablets, debossed " є343" on one side and a full and partial bisect on the other side. They are available in bottles of 100 tablets (NDC 42806-343-01) 20 mg: pink, round biconvex tablets, debossed “ є ” above “ 344 ” on one side and quadrisect on the other side. They are available in bottles of 100 tablets (NDC 42806-344-01). 30 mg: pink, round flat-faced beveled edge tablets, debossed "є" above "345" on one side and quadrisect on the other side. They are available in bottles of 100 tablets (NDC 42806-345-01). Dispense in a tightly-closed, light-resistant container as defined in the USP, with a child-resistant closure, as required. Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature]. Dispense with Medication Guide available at: www.epic-pharma.com/medguide/Dextroamp-Saccharate-Amp-Asparate-Dextroamp-Sulfate-Amp-Sulfate-Tablet.pdf Distributed by: Epic Pharma, LLC Laurelton, NY 11413 Rev.09/2023 MF339REV09/2023 OE2729
More pills like ROUND logo 341