pantoprazole sodium tablet delayed release
1 INDICATIONS AND USAGE Pantoprazole Sodium Delayed-Release Tablets, USP are indicated for: Pantoprazole is a proton pump inhibitor (PPI) indicated for the following: Short-Term Treatment of Erosive Esophagitis Associated with Gastroesophageal Reflux Disease (GERD) ( 1.1 ) Maintenance of Healing of Erosive Esophagitis ( 1.2 ) Pathological Hypersecretory Conditions Including Zollinger-Ellison (ZE) Syndrome ( 1.3 ) 1.1 Short-Term Treatment of Erosive Esophagitis Associated With Gastroesophageal Reflux Disease (GERD) Pantoprazole is indicated in adults and pediatric patients five years of age and older for the short-term treatment (up to 8 weeks) in the healing and symptomatic relief of erosive esophagitis (EE). For those adult patients who have not healed after 8 weeks of treatment, an additional 8-week course of Pantoprazole may be considered. Safety of treatment beyond 8 weeks in pediatric patients has not been established. 1.2 Maintenance of Healing of Erosive Esophagitis Pantoprazole is indicated for maintenance of healing of EE and reduction in relapse rates of daytime and nighttime heartburn symptoms in adult patients with GERD. Controlled studies did not extend beyond 12 months. 1.3 Pathological Hypersecretory Conditions Including Zollinger-Ellison (ZE) Syndrome Pantoprazole is indicated for the long-term treatment of pathological hypersecretory conditions, including Zollinger-Ellison (ZE) Syndrome.
lannett company, inc.
Related Pills
pantoprazole 40 MG Delayed Release Oral Tablet
Kremers Urban Pharmaceuticals Inc.
pantoprazole sodium tablet delayed release
lannett company, inc.
Metformin 1 g sitagliptin 100 mg Janumet
Merck Sharp & Dohme Corp.
Allegra 180 MG Oral Tablet
Chattem, Inc.
Fexofenadine hydrochloride 180 MG Oral Tablet
Physicians Total Care, Inc.
tizanidine 4 MG as tizanidine hydrochloride 4.58 MG Oral Tablet
American Health Packaging
liothyronine 5 ug
sigmapharm laboratories, llc
tizanidine 4 mg
American Health Packaging
DISCLAIMER:
"This tool does not provide medical advice, and is for informational and educational purposes only, and is not a substitute for professional medical advice, treatment or diagnosis. Call your doctor to receive medical advice. If you think you may have a medical emergency, please dial 911."
"Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. We may limit or otherwise restrict your access to the API in line with our Terms of Service."
"This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product."
PillSync may earn a commission via links on our site









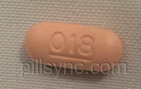




16 HOW SUPPLIED/STORAGE AND HANDLING How Supplied
PANTOPRAZOLE SODIUM Delayed-Release Tablets, USP are supplied as 40 mg white to off-white, oval-shaped coated tablet, debossed with "17" on one side and are available as follows: NDC 62175-617-46, bottles of 90 NDC 62175-617-43, bottles of 1000
PANTOPRAZOLE SODIUM Delayed-Release Tablets, USP are supplied as 20 mg white to off-white, oval-shaped coated tablet, imprinted in black with “18” on one side and are available as follows: NDC 62175-618-46, bottles of 90 NDC 62175-618-43, bottles of 1000 Storage Store
PANTOPRAZOLE SODIUM Delayed-Release Tablets, USP at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature] .
More pills like OVAL 18